Aardvark
Orycteropus afer

2025 Red list status
Near Threatened
Regional Population Trend
Declining
Change compared
to 2016
Uplisted
Overview
Orycteropus afer – (Pallas, 1766)
ANIMALIA – CHORDATA – MAMMALIA – TUBULIDENTATA – ORYCTEROPODIDAE – Orycteropus – afer
Common Names: Aardvark, Antbear (English), Aardvark, Erdvark, Erdmannetjie (Afrikaans), Sambane (Ndebele, Swati, Zulu), Thakadu (Sesotho, Setswana), Xiyambana (Shangaan), S-ámbane (Swazi), Thagalu (Tshivenda), Ihodi (Xhosa), Xomboni (Xitsonga), Erdferkel (German), Oryctérope (French)
Synonyms: Myrmecophaga afra Pallas, 1766; Myrmecophaga capensis Gmelin, 1788
Taxonomic Note:
Aardvarks used to be considered congeneric with South American anteaters (Myrmecophaga sp.) until they were classified as their own genus: Orycteropus (Lehmann 2009). After 1872, Aardvarks were further classified as their own order: Tubulidentata. This order was long considered to be closely related to the Xenarthrans and the pangolins in the now obsolete clade “Edentata” (Lehmann 2007). At the beginning of the 20th century, Aardvarks were considered basal “ungulates” (Le Gros Clark & Sonntag 1926). By the middle of the century, the seven known species of Aardvarks were merged into a single species Orycteropus afer (Shoshani et al. 1988). Since then, the order Tubulidentata is the only order of mammals to be represented by a single living species. To date, 18 subspecies of O. afer have been described (Meester 1971), although their validity is doubtful and studies in this regard are ongoing (Pohlova et al. 2014). At the turn of the millennium, molecular phylogenetic analyses integrated the Aardvark into the super-cohort Afrotheria, alongside with Paenungulata (including Proboscideans, i.e., elephants, Sirenians, i.e., sea-cows, Hyracoideans, i.e., dassies), and Afroinsectivora (such as elephant shrews or sengis, tenrecs, otter shrews, and golden moles; Waddell et al. 2001; Tabuce et al. 2008).
Red List Status: NT – Near-Threatened, A3c (IUCN 2012, version 3.1)
Red list assessment
Assessors: Weyer, N.M.1, 2, 3, Power, R.J. 4 , Taylor, W.A.1, 5
Reviewer: Lehmann, T.1, 6
Contributors: Fuller, A.2, Patel, T.7 & da Silva, J.8
Previous Assessors: Taylor, W.A., Cilliers, S., Meyer, L.C.R. & Wilson, A.-L.
Previous Contributor: Child, M.F.
Institutions: 1IUCN SSC Afrotheria Specialist Group (Aardvark Section); 2Wildlife Conservation Physiology Lab, School of Physiology, University of the Witwatersrand, Johannesburg, South Africa; 3IUCN Centre for Species Survival (CSS) Small Mammals, Alpenzoo Innsbruck-Tirol, Austria; 4Department of Economic Development, Environment, Conservation and Tourism, North West Provincial Government; 5Panthera, Steenberg Office Park, Tokai, Cape Town, 7945; 6Senckenberg Research Institute and Natural History Museum Frankfurt, Senckenberganlage 25, 60325 Frankfurt am Main, Germany; 7Conservation Planning and Science Unit, Endangered Wildlife Trust, South Africa; 8South African National Biodiversity Institute, Kirstenbosch Research Centre, Cape Town, 8000, South Africa.
Assessment Rationale
This regional assessment provides a justification for uplisting the Aardvark (Orycteropus afer) from “Least Concern” to “Near Threatened” in the assessment region (South Africa, Eswatini and Lesotho) based on IUCN Red List Criterion A3c.
Although widespread within the assessment region, Aardvarks are nowhere common and generally occur at low densities (for example, a study in the Karoo estimated a density of ca. 8 animals per 10 km2). The species is globally classified as Least Concern by the International Union for Conservation of Nature (IUCN) Red List (Taylor & Lehmann 2015), and was last assessed in this assessment’s region in 2016 (Taylor et al. 2016). Aardvarks are sometimes considered rare because of their elusive behaviour, but not necessarily because of low numbers, as no population size data are currently available. The reproductive rate of Aardvarks is low (maximum one offspring per year), making populations potentially vulnerable to disturbances that cause declines. The emerging threat of climate change for instance is likely exacerbating the already existing threat to Aardvarks particularly in the arid and also in the more mesic parts of their distribution within the assessment area. While Aardvarks are able to survive in semi-arid regions, they are strongly affected by droughts in these environments. Aardvarks do not occur in deserts. The ongoing climate-change related desertification of habitats in the assessment region will likely reduce large parts of suitable habitat over the next three generations of Aardvarks, reducing their extent of occurrence (EOO). Climate-change attributed drought also has an impact on Aardvark food resources. Resource scarcity reportedly caused physiological challenges, starvation and mass mortalities of Aardvarks, thereby likely causing local population to decline in some areas in the past decade and in future, and possibly extirpation of Aardvark populations in the assessment region. In summary, environmental stressors, such as drought- or flood-induced lack of resources and increased heat load brought about by climate change, pose a greater threat to Aardvarks than previously considered.
Furthermore, the killing of Aardvarks by humans for wild meat and traditional medicine or retributional persecution for (allegedly) damaging fences and croplands likely exacerbate negative effects on Aardvark populations. As a result of this human-wildlife-conflict, Aardvark numbers are likely reduced in areas where their habitat is altered by human activities or where human density is high. South Africa’s human population grew by 19.8 % between 2011 and 2022 and is expected to increase going into the future. The concomitant increase in human activities could negatively impact Aardvark populations further. While quantifying these effects on Aardvarks is difficult, it is nevertheless important to attempt it using different methods, including field observations and citizen science.
Counting Aardvarks in their natural habitats is also difficult and no systematic approach has yet been developed to monitor population numbers. As a result, no assessment can currently be made about population trends based on direct observations. However, future population trends can be inferred from knowledge on the impacts of climate change on the survival of Aardvark individuals.
Following Section 12 of the IUCN Red List Guidelines (Version 16; IUCN 2024), the following rationale for uplisting the Aardvark from Least Concern to Near Threatened is used:
- The underlying mechanism of the impact of climate change on Aardvarks in the region is a reduction in fecundity and survival caused by increases in aridity, heat, and likelihood of severe drought conditions that lead to population reductions through reduced numbers of mature individuals. Recent studies in the Kalahari have shown that climate change (especially through drought) is causing declines in Aardvark prey (ant and termite) availability, which leads to poor body condition in Aardvarks, cessation of reproduction, and even mortalities during prolonged or extreme drought conditions.
- The Aardvark’s current EOO covers nearly the entire assessment region ((Taylor & Lehmann 2015; see also Figure 1). Climate change and ecosystem zone models observe and predict large-scale increases in heat and aridity as well as an expansion of aridification in at least 40 % of the assessment region and therefore the Aardvark’s EOO within the next few decades7, 8. It is, therefore, likely and plausible that at least 30 % of the available habitat of Aardvarks in the region will be lost to the species over its next three generations.
- The status of the Aardvark in the assessment region is, therefore, uplisted from Least Concern to Near Threatened under IUCN Red List Criterion A3c, because it is close to qualifying for the category Vulnerable A3c, or is likely to do so in the near future: A population reduction of ≥30 % is projected (inferred) to be met within three generations, based on (indirect evidence of) a decline in EOO and quality of habitat.
The Aardvark appears to be under greater threat than previously considered in any population assessment. Furthermore, threats to Aardvarks might have cumulative impacts on their populations, for example, if the threats overlap geographically. Long-term systematic monitoring of subpopulation trends is needed to assess the impact of these threats. Conversion from livestock to wildlife ranching may counteract habitat loss for this species and sustain the ant and termite populations on which this species depends for food and water, if persecution is abolished. Similarly, the effectiveness of interventions to reduce the perceived need for persecution, such as swing-gates and tyres to prevent Aardvarks from digging holes underneath fences, should be tested. Nevertheless, halting the impacts of climate change on environments it inhabits is vital for ensuring the ongoing presence of this species and thus its ability to continue delivering its ecosystem services.
Reasons for Change
Reason(s) for Change in Red List Category from the Previous Assessment:
The Aardvark used to be classified as Least Concern1 in the assessment area (Taylor et al. 2016) and globally (Taylor & Lehmann 2015) due to it being widespread and regularly seen throughout its range, despite unknown population numbers. However, the perception that populations are thriving has shifted in recent years among Aardvark researchers, following reported impacts of climate-change attributable drought events in semi-arid parts of the assessment region on Aardvarks, justifying an uplisting. Here, we provide a rationale for uplisting the Aardvark to Near-Threatened2 within the assessment region.
Drought-related food scarcity in a semi-arid part of the assessment region reportedly impacted the Aardvark population in two ways. Firstly, Aardvarks died from starvation caused by drought-related resource scarcity and resulting starvation caused a population size reduction3 through death of individuals. Second, the number of mature4 individuals among the remaining Aardvarks declined, as survivors did not reproduce under prevailing resource-scarcity and starvation, but only after they had recovered many months later (i.e., causing a population size reduction by reducing the number of mature, i.e., countable individuals).
The Aardvark’s extent of occurrence (EOO; Taylor & Lehmann 2015;) covers nearly the entire assessment region and is thus affected by the observed and projected climate change impacts in Southern Africa5. The current hot and dry conditions of the Kalahari appear to bring the Aardvark’s survival capacity to its limits already (Rey et al. 2017; Weyer 2018; Weyer et al. 2020a); with the projected further increases in heat and aridity affecting large parts of the assessment region, further population impacts are to be expected within the next three generations of Aardvarks in at least 20–30 % of their current distribution. In addition, the current conditions prevailing in the Kalahari are expected to expand to further regions of the assessment area within the next three generations of Aardvarks7, 8, making them potentially uninhabitable to the species, and certainly posing severe survival limitations. Therefore, the Aardvark is close to meeting the Vulnerable-criterion A3c5, and is therefore to be categorised as Near-Threatened.
In addition to this main rationale for our uplisting recommendation, we wish to provide the following information, which may strengthen our argument and be of relevance to follow-up assessments of Aardvarks in the region.
During and after a climate-change related drought, starving Aardvarks, all of which were (by chance) females, were in below-average body condition and did not reproduce in the Kalahari over a 3-year study period (Weyer pers. obs.). Only one Aardvark female, which was in good condition in a non-drought year, produced one offspring (Weyer 2018), but no others did, even after the drought conditions had ceased. Therefore, it is assumed that Aardvarks in poor body condition, as caused by drought-induced food scarcity (Weyer 2018), have limited capacity to reproduce. Aardvarks took long (several months) to recover body condition from starvation once environmental conditions improved. During the recovery time, the Aardvarks also did not reproduce. Therefore, during and after droughts, the number of individuals considered “mature” in the sense of IUCN Red List criteria (i.e., reproductive) was strongly reduced and is likely to decrease again as droughts continue to occur, with increased frequency, duration and severity, in the assessment region.
The estimated generation length6 of Aardvarks in the wild is 9.9 years (Pacifici et al. 2013). Zoo records show female Aardvarks giving birth at 2–3 years age (Shoshani et al. 1998), but there are no data from the wild. However, if environmental factors such as drought and related starvation inhibit the reproduction of Aardvarks, as was observed in the Kalahari, the onset of the reproductive age (or onset of maturity) could be delayed and thus generation length may be extended, in addition to a decline in mature individuals.
In populations reduced by events such as droughts (see Weyer 2018; Weyer et al. 2020a; Rey 2017), the reduction of the number of reproductive individuals combined with an extension of generation length may become particularly problematic for a population’s recovery rate in time for the next drought, as drought frequency is already increasing in semi-arid southern Africa. What is more, is that these conditions will likely impact herbivorous insects, notably the termite (but also ant) prey of Aardvarks, on which they depend strongly in some regions. If these social insects are affected by drought, their colonies can take years to recover even once drought conditions have passed, further exacerbating the conditions for Aardvarks (Nel & Hewitt 1969; Phakoago 2024). Termite colonies in semi-arid habitats of the assessment region are less likely to recover from drought-related crashes than ant colonies (Phakoago 2024), which could become problematic for Aardvarks in regions where they depend more on termites than on ants, such as in the Kalahari semi-desert (Weyer 2018).
In a nutshell: Aardvarks were observed to be killed by climate-change related drought impacts, and surviving ones were in poor condition and did not reproduce, i.e., number of mature individuals was inferred to have declined strongly. Aardvarks reproduce slowly and have a long generation length. They take time to recover from starvation if they survive, but the time between drought events might be too short for that already and more so under progressing climate change and aridification.
1The category Least Concern is applied to taxa that do not qualify (and are not close to qualifying) as Threatened or Near Threatened. It is important to emphasise that “Least Concern” simply means that, in terms of extinction risk, these species are of lesser concern than species in other threat categories. It does not imply that these species are of no conservation concern.”
2A taxon is Near Threatened when it has been evaluated against the criteria but does not qualify for Critically Endangered, Endangered or Vulnerable now, but is close to qualifying for or is likely to qualify for a threatened category in the near future.
3A reduction is a decline in the number of mature individuals of at least the amount (%) stated under the criterion over the time period (years) specified, although the decline need not be continuing.
4The number of mature individuals is the number of individuals known, estimated or inferred to be capable of reproduction. It is relevant for IUCN criteria A-D.
5Population size reduction measured over the longer of 10 years or 3 generations is projected, inferred or suspected to be met in the future (up to a maximum of 100 years) based on a decline in extent of occurrence (EOO) and/or habitat quality; a decline in area of occupancy (AOO) needs to be assessed
6Generation length is the average age of parents of the current cohort (i.e., newborn individuals in the population). Generation length therefore reflects the turnover rate of breeding individuals in a population. Generation length is greater than the age at first breeding and less than the age of the oldest breeding individual, except in taxa that breed only once. It affects IUCN criteria A, C1 and E.
Red List Index
Red List Index: Uplisted
Recommended citation: Weyer NM, Power RJ & Taylor WA. 2025. A conservation assessment of Orycteropus afer. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Regional Distribution and occurrence
Global
The Aardvark is widely distributed in Africa south of the Sahara, including from Senegal to Ethiopia and Kenya to South Africa; it is also present in the Congo Basin, although its distribution in West African rainforests is poorly known; and it is absent from the Sahara and Namib Deserts (Taylor 2013). In southern Africa, the Aardvark is widespread in Botswana, Eswatini, Mozambique, Namibia (except the Namib Desert), South Africa, and Zimbabwe (Monadjem 1998; Skinner & Chimimba 2005). The distribution of the Aardvark is largely determined by the distribution of suitable ant and termite prey species and availability of soils conducive to digging burrows, and by deserts. The species may possibly be perceived as disappearing from countries where wild meat trade is considered a problem (Koutchédi et al. 2023), and it certainly experiences habitat loss locally due to agriculture and human expansion, as well as through desertification. Although its geographic range has likely not changed in recent times, no efforts have been undertaken to measure range contractions or expansions. However, the Aardvark’s range may decline with climate change related aridification and desertification of habitats it currently inhabits.
Within the assessment region:
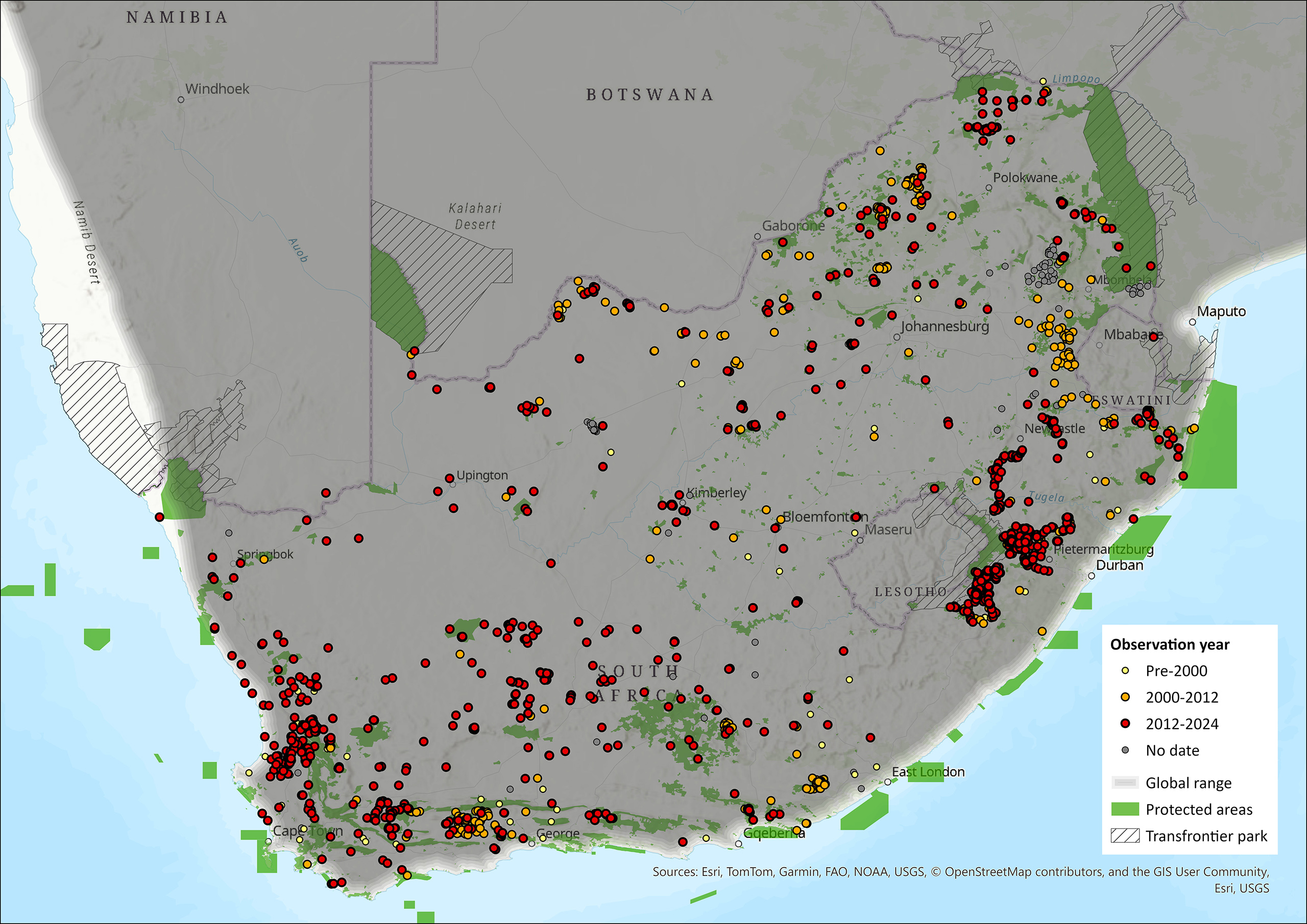
The Aardvark’s extent of occurrence (EOO, Taylor & Lehmann 2015; see also Figure 1) covers nearly the entire assessment region except high mountain ranges and extremely arid Namib Desert stretches along the South African West Coast, and can thus be estimated at ~1,268,756 km².
Figure 1. Distribution records for Aardvark (Orycteropus afer) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
In South Africa (area size ~1,221,037 km²), Aardvarks occur almost throughout the entire country, and sightings indicate presence in all provinces except for small parts of the Western and Northern Cape provinces (Haussmann et al. 2018; Epps et al. 2021; Knöthig 2005; Makwati et al. 2024; Melton & Daniels 1986; Power et al. 2019; Rey et al. 2017; Taylor 1998; Lindsey 1999; Phakoago et al. 2024; Taylor et al. 2002; Taylor & Skinner 2003; Van Aarde et al. 1992; Weyer 2018; Weyer et al. 2020a). A recent study used formally collected records to map Aardvark distribution and used publicly available social media footage (680 photographs and videos of Aardvarks over a ten-year period, 2010–2019, across southern Africa) to gather information on their distribution and ecology (Phakoago et al. 2024). The study revealed that in South Africa, Aardvarks were seen during all seasons and in all provinces, with most sightings in the Northern Cape (34 %; mostly Tswalu Kalahari Reserve), Limpopo (19 %, mostly Kruger National Park), and the Eastern Cape (18 %; Phakoago et al. 2024). Though Aardvarks are present in regions in the Northern Cape that are part of the Kalahari semi-desert, these populations have likely been impacted during mass mortalities caused by recent summer droughts (Rey et al. 2017; Weyer 2018; Phakoago et al 2024), although quantitative data are not available. Aardvarks also occur on human-disturbed habitats, such as farmlands in the Natal Midlands that are grazed or cut. Aardvarks may have gone extinct from the West Coast National Park due to agricultural practices in the region bordering the park (Knöthig 2005). Aardvarks do not occur in the Namib desert part that reaches into South Africa.
In Eswatini (area size ~30,355 km²), Aardvarks are said to occur widely, although no population data exist for the country (Monadjem 1998; Knöthig 2005).
In Lesotho (area size ~17,364 km²), Aardvarks might marginally occur (Lynch 1994), but published records from the country are not available.
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): 0
Elevation Upper Limit (in metres above sea level): potentially up to 3200 (based on aardvark sightings in Bale mountains in Ethiopia (Yalden et al. 1996); no elevation data available for the assessment region)
Depth Lower Limit (in metres below sea level): (NA)
Depth Upper Limit (in metres below sea level): (NA)
Depth Zone: (NA)
Biogeographic Realms
Biogeographic Realm: Afrotropical
Countries of Occurrence
| Country | Presence | Origin | Formerly Bred | Seasonality |
| Angola | Extant | Native | – | – |
| Botswana | Extant | Native | – | – |
| Burkina Faso | Extant | Native | – | – |
| Burundi | Extant | Native | – | – |
| Cameroon | Extant | Native | – | – |
| Central African Republic | Extant | Native | – | – |
| Chad | Extant | Native | – | – |
| Congo | Extant | Native | – | – |
| Congo, The Democratic Republic of the | Extant | Native | – | – |
| Djibouti | Extant | Native | – | – |
| Eritrea | Extant | Native | – | – |
| Eswatini | Extant | Native | – | – |
| Ethiopia | Extant | Native | – | – |
| Gabon | Extant | Native | – | – |
| Gambia | Extant | Native | – | – |
| Ghana | Extant | Native | – | – |
| Guinea-Bissau | Extant | Native | – | – |
| Kenya | Extant | Native | – | – |
| Lesotho | Possibly Extant | Native | – | – |
| Malawi | Extant | Native | – | – |
| Mali | Extant | Native | – | – |
| Mozambique | Extant | Native | – | – |
| Namibia | Extant | Native | – | – |
| Niger | Extant | Native | – | – |
| Nigeria | Extant | Native | – | – |
| Rwanda | Extant | Native | – | – |
| Senegal | Extant | Native | – | – |
| Sierra Leone | Extant | Native | – | – |
| Somalia | Extant | Native | – | – |
| South Africa | Extant | Native | – | – |
| Sudan | Extant | Native | – | – |
| Tanzania, United Republic of | Extant | Native | – | – |
| Uganda | Extant | Native | – | – |
| Zambia | Extant | Native | – | – |
| Zimbabwe | Extant | Native | – | – |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: (NA)
FAO Area Occurrence
FAO Marine Areas: (NA)
Climate change
Understanding the physiological and behavioural limitations of free-living animals is vital for assessing their responses to increased environmental stress resulting from contemporary climate change (Fuller et al. 2014), especially in southern Africa, which is already strongly affected by a climate-change related increase in temperatures, drought, aridification and desertification (Gutiérrez et al. 2021). This section details how the Aardvark has been, and may (continue to) be, directly and indirectly impacted by contemporary and future climate change in the assessment region, thus providing a backdrop for the uplisting to Near-Threatened by this assessment.
Africa has been undergoing large-scale common regional changes, as summarised by the IPCC (Gutiérrez et al. 2021): Mean temperatures and hot extremes have emerged above natural variability, relative to 1850–1900, in all land regions in Africa (high confidence). The rate of surface temperature increase has generally been more rapid in Africa than the global average, with human-induced climate change being the dominant driver (high confidence). Observed increases in hot extremes (including heatwaves) and decreases in cold extremes (including cold waves) are projected to continue throughout the 21st century with additional global warming (high confidence). The frequency and intensity of heavy precipitation events are projected to increase almost everywhere in Africa with additional global warming (high confidence). These changes generally apply to the region encompassed by this assessment7.
The Aardvark’s extent of occurrence (EOO; Taylor & Lehmann 2015;) covers nearly the entire assessment region. It is thus affected by the observed and projected climatic impacts in Southern Africa7 and is part of the West and East Southern Africa regions of the IPCC analyses. Specifically in these regions, decreases in mean precipitation, increases in heavy precipitation and pluvial flooding; increases in aridity, agricultural, meteorological and ecological droughts have already been observed (Gutiérrez et al. 2021). Other studies reported increasing number or frequency of droughts in the more recent decades (Manatsa et al. 2017). Drought frequency has increased significantly on a 24-month scale since the 1970s (Rouault & Richard 2005) and in relation to El Niño events (Manatsa et al. 2008). For example, from the 1970s to 2016, the frequency of decadal droughts at 3- to 12-months scale increased in South Africa (Richard et al. 2001), and on average, they occurred in conjunction with heatwave events (Yuan et al. 2018).
The following regional synthesis projections were made recently by the IPCC for the West and East Southern Africa regions (Gutiérrez et al. 2021): Mean surface temperature is projected to increase (high confidence), following on from an observed upward trend without attribution. Extreme heat is also projected to increase (high confidence), following on from an observed upward trend with high confidence of attribution. Mean precipitation is projected to decrease (high confidence), with a downward trend already reported without attribution. Hydrological droughts will increase (medium confidence). Aridity is projected to increase (high confidence). Agricultural and ecological drought are projected to increase (medium to high confidence), following on from an observed upward trend without attribution. Fire weather conditions and mean wind speed are projected to increase. Other, including even more recent publications, corroborate the trend of increasing intensity, severity and impact of drought on vegetation (see, e.g., Chivangulula et al. 2023; Lawal et al. 2022).
The Kalahari semi-desert at the south-western edge of Aardvark distribution is the hottest and driest environment currently inhabited by Aardvarks (Weyer 2018) and the hot and dry climatic conditions prevailing there are already and will continue to be expanding throughout the assessment area7, 8, with possible severe impacts on habitat suitability for Aardvarks within the next three generations.
Aardvarks are already at the physiological limit in this region: Following a particularly dry and hot summer in 2012/2013 and the subsequent winter of 2013, many Aardvark deaths occurred in a Kalahari study site (Rey et al. 2017; Weyer 2018; Weyer et al. 2020a). In this region, the main prey item of Aardvarks is the Northern Harvester Termite, H. mossambicus, which provides Aardvarks with up to 96 % of their nutrition and water, while the remainder is provided by the Snouted Termite, Trinervitermes trinervoides, the Pugnacious Ant Anoplolepis custodiens, and other ants and termites (Weyer 2018). Harvester termite abundance is correlated with grass availability, which in turn is dependent on rainfall (Weyer 2018). Good rainfall in the wet seasons leads to high vegetation productivity, resulting in high ant and termite abundances, enabling Aardvarks to obtain sufficient prey to cover their minimum daily energy and water requirements and maintain good body condition (Weyer 2018). As for other mammals, records of body temperature and activity patterns provided useful direct indicators of physiological well-being of Aardvarks (Weyer 2018). Irrespective of the season, Aardvarks in good condition remained nocturnal, and maintained a tightly controlled body temperature (35.4–37.2 °C), with a small 24-h amplitude of their body temperature rhythm (~2.5 °C; Weyer 2018). However, during the summer drought 2012/2013, body temperature of Aardvarks fluctuated progressively through the dry summer months, with some body temperatures reaching as low as 25 ˚C before death, likely due to starvation (Rey et al. 2017; Weyer 2018). During this summer drought, Aardvarks also relaxed the precision of body temperature regulation (mean 24-h amplitude 2.3±0.4 ˚C). Individuals that subsequently died became increasingly diurnal in the weeks before their deaths. Throughout the following winter, the Aardvarks’ minimum 24-h body temperatures declined, causing exaggerated heterothermy (4.7±1.3 ˚C; range 24.7 to 38.8 ˚C), with one individual’s body temperature varying by as much as 11.7 ˚C within 8 h (Weyer et al. 2020a).
In the Kalahari, under these drought conditions, activity patterns shifted from the normal nocturnal to a diurnal mode (Rey et al. 2017; Weyer 2018). When body temperatures were low, Aardvarks often emerged from burrows during daytime and sometimes basked in the sun to passively gain heat (Weyer 2018). They occasionally even returned to burrows before sunset, resulting in completely diurnal activity (Weyer et al. 2020a). While these shifts in body temperature and activity point at a certain physiological and behavioural flexibility, Aardvarks were unable to compensate for reduced food availability during the summer drought, resulting in starvation and in many cases, eventually, death (Weyer et al. 2020a). A recent study assessing a large set of social media footage reported that Aardvarks throughout southern Africa were seen more often at night, except in winter, and that Aardvarks in poor body condition were more likely to be seen in daylight, while Aardvarks seen at night were in good condition (Phakoago et al. 2024). Being more diurnal may be a suitable mechanism to save energy in winter, when food tends to be scarce and days are milder than nights (Van der Vinne et al. 2015). If, however, this mechanism drives Aardvarks to become diurnal during summer, exposure to daytime heat, especially during drought, may put them at risk of desiccation, especially in water-scarce environments.
In addition to food energy, Aardvarks are likely to cover their water requirements mainly from their prey and are not generally dependent on external water sources to meet their water requirements if sufficient prey is available (Taylor & Skinner 2004; Pietersen et al. 2016; Taylor et al. 2018). However, during periods of prey scarcity, Aardvarks might need to supplement their water intake. Although Aardvarks might drink occasionally, only few records of Aardvarks drinking water in the Karoo had been documented in field studies until recently (Taylor & Skinner 2004; Skinner & Chimimba 2005; Kerley & Tompkins 2017). The study that mined social media data on Aardvarks and Ground Pangolins (Smutsia temminckii) contained the largest-ever data set of drinking for both species, with 32 drinking observations of Aardvarks (Phakoago et al. 2024). This study suggests that both these ant- and termite-dependent species may become more reliant on drinking water when exposed to higher heat loads in summer, and when food is scarce (and hence, water intake through food is reduced; Weyer 2018). Many of the published images of drinking Aardvarks showed individuals in poor body condition (Phakoago et al. 2024; see also images in Kerley & Tompkins 2017), corroborating the suggestion that Aardvarks might become more water-dependent during times of nutritional restriction.
The ability to cover their water requirements from their prey allows Aardvarks to occur in habitats where free-standing water is not readily available, such as the Kalahari semi-desert, if sufficient prey is available (Taylor et al. 2018). However, water access especially in the semi-arid habitats of the Aardvark’s geographic range might become even more limited under climate change than it already is, potentially restricting the distribution of Aardvarks, especially if their prey insect populations are also affected by climate change, in turn possibly making the availability of drinking water more important for Aardvarks than at present (Taylor et al. 2018; Weyer 2018).
Increasing frequency and severity of droughts across southern Africa will result in a decline in vegetation abundance, impacting strongly on termite and ant abundances on which Aardvarks are strongly dependent for nutrition and water (Weyer 2018). Populations of many insect taxa experience lasting impacts of climate change and related extreme events such as drought and rains, and generally, those with limited dispersal capacity (such as colony-forming ants and termites) are at highest risk of extinction from rapid climate change (e.g., Parmesan 2006; Deutsch et al. 2008; Halsch et al. 2021; and references therein). Declines, increased competition, and die-offs of populations of herbivorous ant species in response to drought have occurred within and outside the assessment region, an effect that is exacerbated by ongoing climate change (Couper et al. 2021; Sundaram et al. 2022). Termite activity is temperature dependent and limited by high temperatures (e.g., Ohiagu 1979). Drought impacts on vegetation that termites depend on can cause populations to crash and not recover even once drought conditions have passed (Nel & Hewitt 1969). During a recent study in the Kalahari, higher than average rainfall (2020 and 2021) following a very hot and dry period in 2019 resulted in high grass cover and an increase in ant activity, whereas termite numbers remained low. Nevertheless, Aardvarks preferred termites over ants in their diet over the study period, and consumed predominantly termites of the genus Trinervitermes (45 % of their diet during this study; Phakoago 2024) instead of their staple prey in this habitat, the Northern Harvester Termite H. mossambicus (up to 96 %; Weyer 2018), likely because its populations had not sufficiently recovered to serve as main prey item. Increased heat during climate change will restrict foraging and increase drought intensity and frequency, as well as aridification, in the assessment region. Concomitantly, climate change in the assessment region will likely lead to lower herbivore carrying capacity, possibly further reducing ant and especially termite populations and thus prey availability to the Aardvark over extended periods, limiting Aardvark survival capacity.
Aardvarks might struggle to meet energetic requirements during extremely dry seasons in the arid Kalahari (Weyer et al. 2020a), and in extremely wet seasons in the more mesic Waterberg (Chen et al. 2023). Therefore, Aardvarks may be walking a nutritional tightrope in that both particularly dry seasons and particularly wet seasons appear to negatively affect their prey availability and hence energy intake. In addition to drought, in areas where climate change is predicted to increase the frequency and severity of other extreme events (such as floods), Aardvarks might thus also be at risk of extirpation.
Finally, a recent genetic study suggested that gene flow between Aardvark populations is limited in arid areas. Hence, climate change (i.e., aridification) could increase habitat fragmentation and further limit gene flow for Aardvarks (Epps et al. 2024), although this hypothesis needs to be ground-truthed.
As studies have shown, the current hot and dry conditions of the Kalahari appear to bring the Aardvark’s survival capacity to its limits already (see Sections “Reasons for Change” and “Ecology”); with the ongoing further increases in heat and aridity7, further population impacts are to be expected within the next three generations of Aardvarks. In addition, the current conditions prevailing in the Kalahari are expected to expand to further regions of the assessment area within the next three generations of Aardvarks8, making large parts of their current habitat potentially uninhabitable to the species.
Population information
Current Aardvark population sizes and trends are not quantified. In Southern Africa, due to a lack of systematic surveys, there is no evidence whether population density is either decreasing or increasing significantly in direct response to any physical parameter. However, environmental fluctuations in some of their habitats, such as the Kalahari semi-desert, are likely to induce at least local declines in population size and density, as climate-change related droughts caused mass mortalities of Aardvarks in this dry habitat. Aardvark densities likely vary according to habitat suitability and prey abundance but cannot be directly tied to observed burrow densities as Aardvarks change burrows frequently and an area may contain many abandoned burrows (Taylor 2013; Weyer 2018). Moreover, the number of burrows maintained by an Aardvark in its home range likely differs depending on the soil characteristics in its environment. A single Aardvark may easily generate >100 burrows in its home range in a sandy habitat (Weyer 2018). The only study that estimated Aardvark densities provided estimates of 8 Aardvarks per 10 km² in the Karoo (Taylor & Skinner 2003).
Within the assessment region, the number of mature individuals is unknown but likely exceeds 10,000 individuals, given their extensive distribution. This estimate could possibly represent 10–30 % of the global population, which has not been quantified yet, either.
Their level of population fragmentation is unknown but probably low, because Aardvarks occur inside and outside of protected areas and are therefore able to move rather freely. Fences are not always a barrier to Aardvarks as they are able to burrow underneath them (Schumann et al. 2006). Aardvarks can cover distances of 2.7–8.4 km per night when foraging (van Aarde et al. 1992; Taylor & Skinner 2003; Weyer pers. obs.), thus they may possess dispersal potential. While individuals may cover larger distances, their populations are considered to stay in close proximity of each other (<55 km), contributing to a low level of fragmentation (Epps et al. 2024). In terms of regional population effects, so-called rescue effects, for example through migration of individuals between neighbouring regions, are possible, because much current Aardvark habitat within the assessment region is connected to natural habitat outside of the assessment region, especially in its semi-arid parts. Yet during severe drought conditions in the Kalahari, Aardvarks were not observed to disperse to new or different areas but remained within their home ranges and re-used the same burrows for longer consecutive periods than during non-drought conditions (Weyer 2018; Weyer unpublished data). Therefore, there may be limitations to unsupported dispersal of the species under environmental challenges and climate change.
Reproductive and survival rates of free-living Aardvarks are unknown. Zoo Aardvarks can live up to 14–32 years (Goldman 1986; BBC 2023), although it is unlikely that free-living Aardvarks reach the same age. Although young zoo Aardvarks grow quickly, this does not appear to be the case in the wild (Taylor pers. obs.), where their natural diet is restricted to ants and termites. Female zoo Aardvarks give birth at 2–3 years age (Shoshani et al. 1988), but there are no data from the wild. Estimated generation length of Aardvarks in the wild is 9.9 years (Pacifici et al. 2013).
Wildlife ranching management practices likely preserve natural habitat suitable for Aardvarks if ant and termite populations remained untouched by human activities and unaffected by the impacts of climate change. Moreover, increased trampling of grass pastures by cattle was suggested to create favourable conditions for termites (Whittington-Jones 2006). Termite presence, in turn, may be beneficial: in grassland ecosystems, the benefits of termite presence were considered to outweigh any harmful effects, which tend to be associated with degraded, overgrazed grassland and drought (Adam et al. 2018). Ant and termite presence may even benefit crop yield in arid regions (Evans et al. 2011) and increase the robustness of dryland ecosystems to climate change (Bonachela et al. 2015). Similarly, ants can have beneficial effects, as in semiarid savannas, their nests can serve as drought refugia for grass survival and seed source for recovering grass populations after drought (Nicolai et al. 2008). The increase in area under wildlife ranching that has occurred over the past 40 years (Taylor et al. 2021) has possibly benefited the Aardvark population, but is likely insufficient to stabilise their populations, especially in the light of climate change. Aardvarks may be persecuted by farmers as a damage causing animal on some wildlife ranches or farms because they can dig under fences and dig holes in the ground in which cars may get damaged or cattle get stuck or injured. However, the degree of persecution or its possible impacts on the Aardvark population have not been quantified.
The compiled evidence suggests that Aardvark populations may experience declines, especially in smaller natural or protected areas due to the (possibly cumulative) impacts of climate change, habitat loss, and conflict and coexistence challenges with humans. To corroborate this evaluation quantitatively and improve our understanding of Aardvark population sizes, dynamics, subdivision into subspecies, and suspected local declines, further research and monitoring are needed.
Extreme fluctuations in the number of subpopulations: Possible during extreme events, such as summer drought
Continuing decline in number of subpopulations: (Not specified)
All individuals in one subpopulation: (Not specified)
Number of mature individuals in largest subpopulation: (Not specified)
Number of Subpopulations: Inconclusive
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 100 years: (Not specified)
Population genetics
Historically, Aardvarks used to be subdivided in 18 doubtful subspecies (Lehmann 2007), with the nominotypical subspecies O. a. afer coming from South Africa (Cape of Good Hope). Two further supposed subspecies were found in Southern Africa: O. a. angolensis from southwestern Angola and O. a. albicaudus from Namibia and Botswana.
In terms of phylogenetic relationships, a preliminary genetic analysis on European zoo Aardvarks of known descent showed only an insignificant degree of genetic differentiation between Aardvarks from Namibia and Tanzania, even suggesting the possibility of phylogenetically recent (i.e., during the Quaternary) contact between these Aardvark populations despite their geographic distance of ~1,200 km (Pohlova et al. 2014). Conversely, a recent population genetics study based on 19 microsatellite loci and 253 Aardvark faecal samples from nine protected areas in South Africa detected genetic structures suggesting three regional subpopulations of Aardvarks in South Africa, with some admixture, individuals forming a fourth cluster with membership to gene pools not well represented in South Africa (Epps et al. 2024). The genetic clusters included (1) Kruger National Park, (2) central South Africa (Tankwa Karoo, Roggeveld, Karoo, Camdeboo and Victoria West), (3) western/northern South Africa (Namaqua, Augrabies Falls National Park), and (4) the admixed cluster (Upington, Prieska and Eswatini). Although sampling was restricted to a limited number of protected areas, pronounced clusters were detected. Nevertheless, some anatomical and ethological evidence points toward a more significant distinction between Southern African and central African forest Aardvarks (Pagès 1970; Lehmann 2004), which needs to be confirmed by genetic analyses.
Additional genetic studies incorporating more comprehensive sampling across Southern Africa (for instance on a continuous trail) would help make better-informed conservation decisions in the assessment region. Pending such study, management decisions, such as during rehoming confiscations, rehabilitations, or translocations of individuals from different regions should be well-documented, and weigh both the benefits and the risks.
Habitats and ecology
Home ranges. Aardvarks occur in a broad range of habitats, including the semi-arid Karoo and Kalahari deserts, grasslands, all savannah types, rainforests, woodlands and thickets (Shoshani et al. 1988; Taylor 2013). They are well-adapted to multiple habitats, including semi-arid regions, if sufficient prey is available (Taylor & Skinner 2004). Aardvarks are absent from arid and hyper-arid habitats such as the Namib Desert, and scarce in very rocky terrain that is difficult to dig in, as they require soils amenable to digging for food and that are deep enough to dig burrows. They occur at all altitudes within the assessment region but are not common on steep slopes (Skinner & Chimimba 2005; Taylor 2013). They are also known to occupy farmlands, for example in the Drakensberg Midlands of the KwaZulu-Natal Province (Ramesh & Downs 2015). Mean home range size in the Karoo was estimated at 3.5 km2 for both sexes, with 2–4.7 km2 for males and 4.4–4.6 km2 for females (van Aarde et al. 1992), whereas a subsequent study in the same region estimated 2.4–2.7 km2 for females and 2.1–3 km2 for males (Taylor & Skinner 2003). Aardvarks had similar occurrence patterns in a private protected area (Sanbona Private Wildlife Reserve) and farmland in ecologically similar part of the Karoo in the Western Cape (Woodgate et al. 2023). Home range sizes of Aardvarks in the Kalahari were not published at the time of this assessment, but Aardvarks reportedly changed burrows on a nearly daily basis except during periods of drought-related food scarcity (Weyer 2018). Aardvark home range sizes might potentially have a maximum size of 41 km2 (circular) or 21 km2 (elliptical; based on faecal DNA genotype matching (using faecal DNA from 12 locations in South Africa, two locations in Eswatini and one location in Kenya during 2016–2018; Epps et al. 2024), though this study was not corroborated yet by field observations.
Diet and energy requirements. Aardvarks feed almost exclusively on ants and termites which they obtain by digging. In the South African False Karoo, a semi-arid summer rainfall region, Aardvarks consumed 13 species of ants and two species of termites, and the pugnacious ant Anoplolepis custodiens was the most dominant prey species (Willis et al. 1992). In the Kalahari, a summer rainfall region more arid than the False Karoo, termites (as opposed to ants) were more important dietary items for Aardvarks than any other item in other habitats (Weyer 2018), even when ants seemed more abundant than termites following a decline in the termite population (Phakoago 2024). Termites also dominated the diet of Kalahari Aardvarks by numbers, with an overall contribution of ~74 %, with the Northern Harvester Termite H. mossambicus constituting almost two-thirds of the diet. The remainder consisted of three categories each contributing less than 15 %: (1) Snouted Termite Trinervitermes trinervoides, (2) Pugnacious Ant A. custodiens, and (3) various other ant and termite genera combined. The main prey termites H. mossambicus and T. trinervoides provided ~90 % of dietary energy and water for Aardvarks, mostly attributable to H. mossambicus, while the remaining ~10 % of energy and water intake stemmed from A. custodiens and other ants and small termites (Weyer 2018). During all seasons, H. mossambicus alone accounted for at least 50 % and up to 95 % of energy in Kalahari Aardvark diet, and for more than 75 % of dietary water, indicating a strong dependence of Aardvarks on the availability of this particular prey insect (Weyer 2018). In a more mesic environment, the Waterberg region of South Africa, Aardvarks primarily preyed on ants such as Pheidole sp. and Dorylus sp. in both the wet and dry season. While the large Northern Harvester Termite H. mossambicus only made up a quarter of the Aardvark’s diet by count, it was the primary source of energy in both seasons (~40 %; Chen et al. 2023), resembling the finding that Aardvarks in the Kalahari relied heavily on harvester termites for energy across all seasons (Weyer 2018).
Aardvarks in the assessment region might struggle to meet energetic requirements in the arid Kalahari even during non-drought conditions but also in extremely wet seasons in more mesic habitats, such as the Waterberg of South Africa. The basal metabolic rate of Aardvarks, like that of other myrmecophagous mammals, might be particularly low (0.128 cm3 O2 per gram per hour in one zoo animal with a body mass 48 kg and unknown sex; McNab, 1984; equivalent to a basal expenditure of 2,150 kJ per 24 h for study aardvarks in the Kalahari with a mean body mass of 35 kg, range 29-42 kg; Weyer 2018). An Aardvark’s daily energy expenditure when active (e.g., performing vital tasks such as foraging or digging burrows) might be five times greater (~11,700 kJ per 24 h; equation no. 8 for similarly-sized desert mammals in Nagy et al. 1999). Estimated average energetic input for Kalahari Aardvarks from their prey was ~2,865 kJ in 24 h (Weyer 2018), covering their basal metabolic needs, but only less than a quarter of their energy needs when active, meaning that active, free-living Aardvarks barely met their minimal energetic requirements in the Kalahari under arid but non-drought conditions (Weyer 2018). In the Waterberg region, during a particularly rainy wet season, Aardvarks’ energy intake from prey was similarly low at ~2,382 kJ per 24 h, indicating that Aardvarks may even be energetically challenged even in a mesic environment (Chen et al. 2023). The particularly high rainfall during the wet season of the study likely altered prey availability in the Waterberg, as high rainfall can reduce surface activity and foraging efficiency of both herbivorous ants (Farji-Brener et al. 2018) and termites (Braack 1995; Davies et al. 2015; Geerts et al. 2016; Ohiagu 1979). These studies indicate that already, in different environments in the assessment region, Aardvarks might struggle to meet their total energetic needs from their prey, even though they may meet their basal energetic needs (energy required during resting). Climate and environmental changes as already observed and predicted in the assessment region are likely to negatively impact Aardvark prey ants and termites (see Section “Climate Change”).
Although Aardvarks may sometimes ingest other insects, such as pupae of scarabaeid beetles or grasshoppers (Taylor et al. 2002), and seeds of the so-called “Aardvark-cucumber” (fruit of the Cucumis humifructus plant; Meeuse 1963) were found in some Aardvark scats, it is unknown whether the Aardvarks purposely targeted these items or ingested them by accident. Outside the assessment region (in central Namibia), some Aardvarks were recorded feeding on fruits of C. humifructus, and Aardvark scats in that area contained seeds of the plant (Johnson et al. 2024). Feeding on the fruit was suggested as a way of increasing their water intake in dry habitats (Melton 1976), although this theory has not been confirmed. Based on observations of study Aardvarks in the Nama Karoo and the Kalahari that had limited or no access to free-standing water and hardly ever drank, Aardvarks likely obtain most of their water requirements from their food, although they may drink if water is available (Taylor et al. 2018). Kalahari Aardvarks were not observed drinking during a 3-year study (Weyer pers. obs.). Aardvarks in arid habitats are dependent on their prey insects for water. Declines in prey items may thus strongly limit access of Aardvarks to sufficient nutrition and water, which will become more vital in the face of ongoing climate change.
Behavioural adaptations. Aardvarks excavate burrows in which they rest during the day and escape predators (Taylor & Skinner 2003; Weyer 2018). Aardvarks are generally nocturnal, although they emerge from their burrows out in the afternoon in cold weather or during periods of food scarcity. For example, during droughts, when food was scarce and Aardvarks were in poor body condition, Aardvarks in the Kalahari shifted their activity patterns from night to daytime, presumably to preserve energy while foraging in the warmer times of the day (Weyer et al. 2020a). Such changes in activity indicate below-average body condition, i.e., that Aardvarks are not meeting their energy demands (Weyer 2018). While this behavioural adaptation is likely a helpful energy-saving mechanism in winter, when the days are warmer than the nights, it may be a disadvantage during summer if it leads to exposure to daytime heat (Weyer et al. 2020a). Aardvarks may also bask in the sun to increase or maintain their body temperature in an energy-saving way when resources are scarce, as observed in the Kalahari (Weyer 2018; Weyer et al. in prep.). See also Section “Climate Change”.
Reproduction. Aardvarks are solitary, presumably only coming together occasionally for very short periods, e.g., for mating; however, very little is known about their reproduction and social interactions in the wild. Aardvarks do not display sexual dimorphism, meaning that males and females can only be distinguished through close examination of their genitals (Pocock 1924; Weyer et al. 2020b). Both males and females possess prominent scent glands, which may be mistaken for testicles, regularly leading to Aardvark spoor or individuals in the field misidentified as males although they may well have been females (Weyer et al. 2020b).
Predators. Natural Aardvark predators include Leopards (Panthera pardus), Lions (P. leo), Spotted Hyenas (Crocuta crocuta), Cheetahs (Acinonyx jubatus), African Wild Dogs (Lycaon pictus), and Pythons (Python sebae; Bothma & Le Riche 1984; Kingdon 1997; Radloff & Du Toit 2004; Hayward et al. 2006; Williams et al. 2018). Although Aardvarks were previously considered a somewhat common prey item for Lions in South Africa (Eloff 1973; Power 2002; Roxburgh 2010), only a single study reported Aardvarks as a potential prey item, as revealed in a systematic literature survey of Lion diet across their distribution range (Hayward et al. 2005). A similar analysis for Leopards revealed three studies documenting the Aardvark as a potential prey item (Hayward et al. 2006). A more recent study that analysed publicly available social media footage (680 photographs and videos of Aardvarks over a ten-year period, 2010–2019, in South Africa) revealed that Leopards appear to be the main predator (84 %) of Aardvarks (Phakoago et al. 2024).
Ecosystem services. Aardvarks are considered a keystone species in many areas as their burrows create a micro-habitat that facilitates the existence of many other vertebrate and invertebrate species (Taylor & Skinner 2000, 2001; Cilliers 2002; Whittington-Jones et al. 2011; Pike & Mitchell 2013; Weyer unpublished data). As many as 39 nocturnal and diurnal species (25 mammals, seven birds, six reptiles and one amphibian) have been reported to use Aardvark burrows as short- or long-term shelter from heat, cold or predators and as a place to rear their young. One notable species that relies on Aardvark burrows is the threatened Blue Swallow (Hirundo atrocaerulea; Evans & Bouwman 2010). Blue Swallows are listed as Critically Endangered in the assessment area (Evans 2015) and as Vulnerable globally (Birdlife 2019), and nest in mist-belt grasslands and construct cup-shaped nests in Aardvark burrows (Wakelin & Hill 2007; Wakelin et al. 2013). A loss of the Aardvark in areas where Blue Swallows occur may have dire consequences for the survival of this endangered bird. Furthermore, various animal species, such as Aardwolves (Proteles cristatus), Ant-eating Chats (Myrmecocichla formicivora) or Clapper Larks (Mirafra apiata) are known to follow Aardvarks and use their fresh feeding pits to access ants or termites, especially during winter, when resources tend to be scarce, and when aardvarks sometimes forage during daylight (Taylor & Skinner 2000, 2001; Vernon & Dean 1988; Weyer pers. obs.).
Aardvarks carry a wide diversity of ectoparasites such as ticks, lice, fleas and flies, and endoparasites including flagellates, amoeba, roundworms, and others (Shoshani et al. 1988). Through their burrows, they also indirectly provide a habitat for argasid tick species, which may be negatively impacted if Aardvarks decline (Nuttall 2022). These ticks are nidicolous, spending their entire life cycle within nests, burrows, or caves, where they have ready access to a bloodmeal. As an example, the African Hut Tampan (Ornithodoros moubata) is a known vector of African swine fever. These ticks live in animal burrows, including those inhabited by Warthogs (Phacochoerus africanus), where they feed on the blood of young warthogs, which rely on their burrow for shelter (Cumming 1975). The availability of burrows for Warthogs is dependent on the Aardvarks that excavate them, and in turn, loss of Aardvarks might thus also adversely impact tick populations that depend on their burrows in some ways, and thereby affect the prevalence of African swine fever virus infections (Bastos & Boinas 2021).
Aardvarks also have a unique functional role in bioturbation and in affecting the plant composition in their natural habitats. By excavating feeding pits that can catch water and provide wind shelter and soil nutrients, Aardvarks provide seed sinks and germination hotspots for various plant species and likely facilitate the establishment of perennial grasses (Chambers & MacMahon 1994; Dean & Milton 1991; Martin 2017; Whitford & Kay 1999). Moreover, abandoned Aardvark burrows provide soil conditions beneficial for seedling survival, allowing the establishment of more species than in undisturbed soils (Haussmann et al. 2018). Through these effects, Aardvarks contribute to floral diversity within the assessment region and potentially across much of their range, with cascading effects such as improved grazing potential of shrubland.
Aardvark extirpation, especially in the more arid parts of their distribution, will likely have severe cascading effects on the numerous animals co-using Aardvark burrows (Haussmann et al. 2018; Weyer et al. 2020a). At the same time, Aardvark burrows will likely become increasingly important for animals seeking refuge from temperature extremes (Pike & Mitchell 2013). Furthermore, with climate change, the frequency of wildfires in regions that are already hot, dry and water scarce will very likely increase (Engelbrecht et al. 2024), making Aardvark burrows potential faunal refuges from fires, as was reported for other burrowing mammals (Linley et al. 2024; Robinson et al. 2013). Their dig sites will also possibly become more relevant plant germination hotspots. Preserving this keystone species and thereby its important ecosystem services is thus especially relevant in the face of ongoing climate change.
Due to their various ecosystem services, the presence of this species is important for wider biodiversity and ecosystem functionality. Further data, notably on population size and density estimates, across their range are required to better assess their populations. This species should be reassessed when such data are available, as it may then even qualify for further uplisting to Vulnerable status in the assessment region in the future.
Cultural services
Throughout its range in Africa, the Aardvark is connected to a wide range of cultural services, for example, hunting them for meat, skin and bones, using body parts for traditional medicines and charms. Various mysticisms and traditional stories revolve around the Aardvark throughout its range, and it has even been depicted in Khoisan cave rock art paintings of up to 6,000 years of age in Zambia (Knöthig 2005).
In the assessment area, according to a written survey, the Setswana and Bisa reportedly use Aardvarks to create potions for luck, and the Griqua, Xhosa and Zulu people use Aardvark body parts for traditional medicine (‘muthi’; Knöthig 2005). The Setswana reportedly see a mystic relationship between humans and Aardvarks (totem) and believe that an Aardvark ‘never looks you in the eye’ and it would be a bad omen if it did (Knöthig 2005). In a more recent study in KwaZulu-Natal, South Africa, surveys indicated that Aardvarks were hunted for meat and to obtain body parts for household use or sale to traditional healers (Makwati et al. 2024). Based on ancient beliefs among traditional healers and their customers that Aardvark body parts possess medicinal or magical value, their claws, teeth, skin, and other parts are used to produce traditional medicine for various purposes including illness prevention, good luck charms, magical crop fertiliser, or poison (Kingdon 1971; Knöthig 2005; Lindsey et al. 2013; Makwati et al. 2024; Melton 1976; Whiting et al. 2011). In addition, Aardvarks are hunted for wild meat both for supplementing household protein and for economic gain (Koutchédi 2022; Taylor et al. 2016). Since their meat is considered a delicacy in KwaZulu-Natal, Aardvarks have been increasingly targeted in hunting activities (Makwati et al. 2024). Unsustainable killing, taking and trade of Aardvarks for traditional medicine, food, trade and recreation likely reduces their population numbers, thereby not only impacting their ecosystem services, but also causing a loss of cultural heritage and traditional knowledge to local and indigenous communities for which they are culturally significant. See also the Sections on Use and Trade, and on Threats.
IUCN Habitats Classification Scheme
| Habitat | Season | Suitability | Major Importance? |
| 1.5. Forest -> Forest – Subtropical/Tropical Dry | – | Suitable | – |
| 1.6. Forest -> Forest – Subtropical/Tropical Moist Lowland | – | Suitable | – |
| 1.9. Forest -> Forest – Subtropical/Tropical Moist Montane | – | Suitable | – |
| 2.1. Savanna -> Savanna – Dry | – | Suitable | – |
| 2.2. Savanna -> Savanna – Moist | – | Suitable | – |
| 3.5. Shrubland -> Shrubland – Subtropical/Tropical Dry | – | Suitable | – |
| 3.8. Shrubland -> Shrubland – Mediterranean-type Shrubby Vegetation | – | Suitable | – |
| 4.4. Grassland -> Grassland – Temperate | – | Suitable | – |
| 4.5. Grassland -> Grassland – Subtropical/Tropical Dry | – | Suitable | – |
Life History
Life history parameters below are based on zoo records and Aardvark life history is likely to differ between zoos and in the wild.
Generation Length: 9.9 years (Pacifici et al. 2013)
Age at Maturity: Female or unspecified: 2–3 years (in zoos; Shoshani et al. 1998)
Age at Maturity: male: (Not specified)
Size at Maturity (unspecified or mixed sex): Size of Aardvarks across their range varies strongly in the literature, with total body length 1,400–2,200 mm; tail length 443–710 mm; length of hind foot: 225–280 mm; ear length: 130–240; shoulder height: 600–650 mm; and body mass: 40–100 kg (Shoshani et al. 1988 and references therein). In the assessment area, study Aardvarks had a mean body mass of 35 kg within a range of 29–42 kg in the Kalahari (Weyer 2018) and a mean body mass of 37.5 kg in the Karoo (Taylor 1998).
Longevity: 14–32 years (in zoos)
Average Reproductive Age: (Not specified)
Maximum Size (in cm): (Not specified)
Size at Birth (in cm): Total length of ~55 cm (Haltenorth & Diller 1977). Body mass of 1,200–1,400 g (Zoo Arnhem) and 1,300–1,900 g (Zoo Frankfurt; Parys et al. 2012).
Gestation Time: 243 days (range = 235–258, n = 6; Taylor 2005)
Reproductive Periodicity: Unknown in the field. All year round in zoos (Parys et al. 2012)
Average Annual Fecundity or Litter Size: 1 per litter
Natural Mortality: (Not specified)
Does the species lay eggs? No
Does the species give birth to live young? Yes
Does the species exhibit parthenogenesis? No
Does the species have a free-living larval stage? No
Does the species require water for breeding? No
Movement Patterns
Movement Patterns: non-migratory; home range fidelitous
Congregatory: No
Systems
System: Terrestrial
General Use and Trade Information
In many parts of their distribution, Aardvarks are hunted for wild meat both for supplementing household protein and for economic gain, and for use in traditional medicine.
In the assessment region, Aardvarks are hunted and killed for meat, curios (skin, claws and teeth), traditional medicinal purposes, and in some cases for trophies (Simelane & Kerley 1998; Whiting et al. 2011; Nieman et al. 2019; Klarmann 2025). Aardvarks have been recorded in traditional medicine markets in the Eastern Cape, Western Cape and Gauteng provinces (Simelane & Kerley 1998; Whiting et al. 2011; Nieman et al. 2019). In the Western Cape, Aardvark skin, bones and oil are traditionally used to protect against evil spirits (Nieman et al. 2019) and sell for 400–2000 South African Rand (equivalent to ca. 30–150 USD, conversion rate at time of publication; Nieman 2018). Aardvark body part trade might be ongoing throughout a greater region within the assessment region. Aardvarks in KwaZulu-Natal were reportedly hunted for subsistence and sale of body parts to traditional medicine markets up to hundreds of kilometres from the site of taking (Makwati et al. 2024), and in the Karoo, Aardvark body parts were owned by a large proportion of the community despite their scarcity in the region, pointing at trade over greater distances (Nieman 2018). Recently, it has become evident through statistics by the Department of Forestry, Fisheries, and the Environment (DFFE) that at least six Aardvarks were hunted for trophies in South Africa in 2023 (Klarmann 2025). While this activity at present does not appear to affect the population in the assessment region, hunting these nocturnal animals was presented by members of the hunting community as an exciting challenge, and it should thus be monitored.
Outside the assessment area, Aardvarks are also reportedly hunted for meat, curios (skin, claws and teeth), and traditional medicinal purposes (e.g., Carpaneto & Germi 1989; Koutchédi & Nobimè 2021; Koutchédi et al. 2023; and others).
No international trade issues or commercial interests are known to date. The species was first listed in 1975, but then deleted in 1993 from the Appendix II of the Convention on International Trade in Endangered Species of Wild Fauna and Flora (CITES).
See also the Sections “Cultural Services” and “Threats”.
| Subsistence: | Rationale: | Local Commercial: | Further detail including information on economic value if available: |
| Yes | Taken for wild meat, curios and medicine in some areas. | Yes | Sold in unregulated markets within and beyond the assessment region. |
National Commercial Value: No
International Commercial Value: No
| End Use | Subsistence | National | International | Other (please specify) |
| 1. Food – human | true | true | true | within and beyond the assessment region. |
| 2. Curios – human | True | True | Possible | within and beyond the assessment region. |
| 3. Traditional medicine | True | True | True | within and beyond the assessment region. |
Is there harvest from captive/cultivated sources of this species? No
Harvest Trend Comments: possibly increasing in some parts of the assessment region (Makwati et al. 2024).
Threats
The existing threats to the species have not been quantified, e.g., in terms of population decline. In an interview survey across various protected areas across its range, i.e., sub-Saharan Africa including the assessment region, preliminary results point at the Aardvark being perceived as threatened through (1) illegal wild meat hunting, (2) climate change, (3) agriculture, (4) logging activities, (5) road kill and (6) human-wildlife conflict (Koutchédi et al. 2023), though these perceptions have not been verified through on-the-ground observations. Local declines are possible due to (cumulative) impacts of habitat loss from agricultural and human settlement expansion and associated subsistence hunting and persecution, and contemporary climate change represents an emerging threat.
Within the assessment region, the following threats (in order of severity) affect the Aardvark:
Climate change: Climate change and climate-change related droughts, mainly through their impact on prey insect availability, pose an emerging and possibly severe threat to Aardvarks in the assessment region during their next three generations and beyond (see also dedicated Section “Climate Change”).
The indirect impact of climate change on termite availability has been identified as a threat to the Aardvark (Rey et al. 2017; Weyer et al. 2020a). Food sources may be affected by increasing aridification of habitat, and could also be impacted by increased precipitation. During a summer drought in 2013, a significant die-off of Aardvarks was observed in the Kalahari (Rey et al. 2017; Weyer 2018; Weyer et al. 2020a). The animals observed as part of an ongoing study at the time of the drought were extremely thin before they died (Weyer 2018). From the temperature and activity data loggers from some of the animals that died and one that survived the drought period, a continuous decline in minimum body temperature during the drought period was observed (Rey et al. 2017; Weyer 2018; Weyer et al. 2020a), which reflects a poor nutritional state in large mammals (Hetem et al. 2016). Moreover, long-term records of the magnitude of heterothermy provided a valuable index of stress within Aardvarks in the Kalahari (Weyer 2018), as previously proposed for free-living, undisturbed large mammals (Fuller et al. 2014). During periods of low food availability and reduced body condition, these Aardvarks also fully shifted their activity patterns from night to day, and foraged during daytime, sometimes starting in the morning (Weyer et al. 2020a). At the time, prey availability was not measured directly; however, based on vegetation surveys and remote sensing (satellite) data, the dry and hot conditions resulted in extremely reduced vegetation growth, which likely drastically reduced the ant and termite prey populations, as is known for various dry habitats (see Section “Climate Change”). Aardvarks in the Kalahari are strongly dependent on their prey, especially the Northern Harvester Termite H. mossambicus for energy and water, so prey insect declines associated with drought and climate change will likely have continuous negative impacts on Aardvark populations.
Outside the assessment region, similar activity shifts and die-offs of Aardvarks have been reported anecdotally during droughts in Namibia in the 1980s (Stuart & Stuart 2001).
Aardvarks likely struggle to meet energetic requirements during extremely dry seasons in the arid Kalahari (Weyer et al. 2020a), and in extremely wet seasons in the mesic Waterberg (Chen et al. 2023). Aardvarks in poor condition are more likely to be active during the day than at night (Weyer et al. 2020a; Phakoago et al. 2024), potentially exposing them to high heat loads, especially under increased temperatures caused by climate change in the assessment region.
Aridification could also have a negative effect on the gene flow between populations, resulting in a more fragmented distribution of Aardvark populations in the assessment region (Epps et al. 2024), although this hypothesis needs to be ground-truthed. More isolated populations with fewer adults capable of reproduction could lead to inbreeding problems and further reduce the adaptability of the species to climate change.
With climate change strongly affecting southern Africa already and in future (Gutiérrez et al. 2021), the hotter and more arid parts of the assessment region will likely see extremely challenging conditions for Aardvarks to survive in, likely leading to Aardvark population decline or local extinctions in some parts of their distribution. Climatic conditions and zones that are currently uninhabitable or at least highly challenging to Aardvark survival have been and will continue to be expanding during their next three generations and beyond7, 8. Increasingly unpredictable precipitation patterns in less arid parts of Aardvark distribution within the assessment region might also generate challenging survival conditions. Climate change is thus likely an even greater threat to Aardvarks than previously considered.
Taking and trade: Hunting of Aardvarks (illegal more so than legal) is considered an ongoing but low-severity threat.
In the assessment region, Aardvarks are used for meat, and their body parts such as skin, claws, teeth, and others are used to produce curios and traditional medicines, either for personal consumption or for sale in illegal/unregulated/traditional medicine markets, for example in the Eastern Cape, Western Cape and Gauteng provinces (Simelane & Kerley 1998; Whiting et al. 2011; Nieman et al. 2019). In the Western Cape, Aardvark skin, bones and oil are traditionally sold to protect against evil spirits (Nieman et al. 2019) at prices ranging between 400 and 2000 South African Rand (equivalent to ca. 30–150 USD as per conversion rate in August 2018; Nieman 2018). Recently, it has become evident through statistics by the Department of Forestry, Fisheries, and the Environment (DFFE) that at least six Aardvarks were hunted for trophies in South Africa in 2023 (Klarmann 2025). While this activity at present does not appear to affect the population in the assessment region, hunting these nocturnal animals was presented by members of the hunting community as an exciting challenge, and it should thus be monitored.
A recent study on perceptions of a rural community about the Aardvark determined different hunting methods of Aardvarks in KwaZulu-Natal (Makwati et al. 2024). Although the study revealed that Aardvarks in KwaZulu-Natal were hunted for meat and to obtain body parts for household use or sale to traditional healers, and could also be killed due to superstitions, it was based on interviews with members of the local community and has not been ground-truthed, for example through field observations or market surveys, so neither extent nor actual impact on aardvarks have yet been verified or quantified.
In the Western Cape, in a survey among traders, Aardvark body parts were found in the possession of 29.4 % community respondents (Nieman 2018). However, even though the species is regionally present (Taylor et al. 2016), live Aardvarks are only infrequently recorded in the area (Nieman 2018), meaning that traded Aardvark body parts might be transported into the area from elsewhere, pointing at the possibility of ongoing Aardvark trade throughout a greater region. Similarly, Aardvarks in KwaZulu-Natal were reportedly hunted for subsistence and sale of body parts to traditional medicine markets up to hundreds of kilometres from the site of taking (Makwati et al. 2024).
Aardvarks can also get caught in wire snare traps intended for wild meat poaching of other species, as reported in the Boland Mountain Complex of the Western Cape (Nieman 2018). Though the level of wild meat poaching is currently not known to be a problematic threat in the assessment region, it is a significant threat to wild species outside the assessment region where it might have a negative impact on the species’ global population (Lindsey et al. 2013). Nevertheless, despite the unknown impact of legal or illegal hunting on the Aardvark population, its sustainability may be questionable because of the regional scarcity of the species (Nieman et al. 2019). See also Sections “Cultural services” and “Use and Trade”.
Agricultural expansion and human-wildlife conflict: This threat is ongoing but considered low-severity.
Currently, Aardvarks lose habitat to agricultural practices, particularly croplands, and are persecuted by farmers because of the damage caused by burrows to dams, fences, roads, mechanical equipment and injury to domestic livestock falling in burrows. Damage to game fences allow valuable game species to escape and allow predators to access game-fenced areas (Schumann et al. 2006; Weise et al. 2014; Rust et al. 2015). For example, in the North West Province, Aardvarks are perceived as problematic by some farmers due to digging holes on roads and blamed for injuries to livestock that have fallen into their holes (Power 2014). Farmers in the North West Province on average rated the Aardvark 1.32 on an intolerance scale from 1 to 5, with two out of 44 farmers perceiving Aardvarks as a cause of damage to livestock that may fall into their holes, as well as to equipment, i.e., vehicles or tractors (Foster et al. 2022).
Conservation
Although the IUCN criteria are designed for global taxon assessments, they can be applied to subsets of global data, especially at regional, national or local levels. In cases where a global category is not the same as a national or regional category for a particular taxon, interactions among sub-units should be carefully considered when planning conservation actions. This case applies to the Aardvark, which is globally classified as Least Concern but is, through this Assessment, considered Near-Threatened in the assessment region due to prevailing regional threats.
Aardvarks are present in many large and well-managed protected areas across the assessment region. No targeted conservation measures are necessary or underway at present, though the need for such interventions should be revisited if further data become available that confirm Aardvark population declines. Conservationists in the assessment region need to make efforts in sharing vital information with farmers, wildlife ranchers and other landowners about the importance of Aardvarks as a keystone species and on potential damage mitigation measures, promote use of wildlife-friendly fences and passageways, and publicise evidence of their effectiveness in reducing persecution rates. For example, in Namibia, swing gates have been an effective alternate passageway for burrowing animals: their installation decreased the number of holes created by Aardvark, as the animals selected swing-gates instead of burrowing (Schumann et al. 2006; Rust et al. 2015). Similarly, installing discarded car tyres into wildlife-proof fences as passageways is a cost-efficient and effective way to reduce damage and facilitate dispersal (Weise et al. 2014). In terms of research, a roadmap for future Aardvark research has been laid out by the IUCN SSC Afrotheria Specialist Group (Aardvark Section), including on Aardvark diet, symbioses and interactions with other species, physiology and reproduction, their conservation status, as well as challenges to Aardvark research (Taylor et al. 2019). In addition, the following research and conservation directions should be considered:
Recommendations for land managers and practitioners in conservation, research and veterinary medicine:
- No management plans are currently necessary, but land managers are encouraged to allow ants and termites to reach natural densities and trial wildlife-friendly fence designs and passageways.
- Practitioners and researchers are cautioned to use correct drug combinations and dosages and implement other precautions when immobilising Aardvarks to minimise risks and enable their smooth recovery (Nel et al. 2000; Rey et al. 2014; Weyer 2018).
Priority topics for research:
- Aardvark population size and trends (e.g., via census or genetic methods) in the assessment region and across its range need to be determined to quantify potential decline rates.
- The full genetic structure of Aardvarks needs to be identified by conducting more comprehensive sampling in the assessment region and across its range, including the identification of possible populations, subspecies, or even new species. For instance, a pilot study initiated by P. Arnold and T. Lehmann in 2024 tries to determine the relatedness of Central African Forest Aardvarks with Southern African ones (supported by IUCN SCC Internal Grant: “Are the forest aardvarks an endangered subspecies (Orycteropus afer subsp.?: genetic evidence from museomics”).
- Effects of main threats to the Aardvark population, especially impacts induced by climate change on Aardvarks, their prey and their habitats, as well as mortalities inflicted by persecution and wild meat taking, need to be quantified.
- Effectiveness of wildlife-friendly fencing in reducing persecution rates needs to be examined.
- Reproductive success and general reproduction behaviour of free-living Aardvarks need to be studied.
Citizen actions and scientific use of social media as a monitoring tool:
- Extraction of social media images recently revealed great potential to obtain supplementary and novel information on the distribution and ecology of elusive species such as Aardvarks and Ground Pangolins (Phakoago et al. 2024). Social media (possibly combined with automated data extraction methods or AI tools) can be used to monitor the species’ activities in various locations simultaneously at low to no cost. Depending on the number of users, social media may even support tracking changing population numbers over time, thereby supporting conservation efforts.
- A large and easily identifiable species as the Aardvark lends itself to being recorded by citizens on virtual museum platforms such as iNaturalist and MammalMAP, although limitations and incorrect labels need to be considered.
- Ongoing collection of images revealing drinking episodes, particularly through camera traps placed at waterholes, would be useful in elucidating the importance of free-standing water in the ecophysiology of the Aardvark, especially considering increasing heat loads and reduced resources under climate change.
Due to the various ecosystem services Aardvarks deliver, the presence of this species is important for wider biodiversity and ecosystem functionality. Once further data, especially on population size and density estimates within and beyond the assessment region as well as across their range are available, the species should be reassessed, as it might then qualify for further uplisting to Vulnerable.
Bibliography
Adam, R.A., Mitchell, J.D. & Van der Westhuizen, M.C. 2018. The role of the harvester termite, Trinervitermes trinervoides (Termitidae: Nasutitermitinae) in semi-arid grassland ecosystems of South Africa: abundance, biomass and grass consumption. African Entomology 26: 36–44. DOI: 10.4001/003.026.0036
Bastos A. & Boinas, F. 2021. Ornithodoros tick vectors and African swine fever virus. In: Nuttall P. (ed) Climate, Ticks and Disease. CABI, Wallingford, pp 405–412. DOI: 10.1079/9781789249637.0058
BBC. 09 June 2023. Colchester Zoo: Europe’s oldest aardvark dies. Accessed on 27 January 2025 at https://www.bbc.com/news/uk-england-essex-65855955
Beck, H.E., Zimmermann, N.E., McVicar, T.R., Vergopolan, N., Berg, A. & Wood, E.F. 2018. Present and future Köppen-Geiger climate classification maps at 1-km resolution. Scientific Data 5, 180214. DOI: 10.1038/sdata.2018.214
Beck, H.E., McVicar, T.R., Vergopolan, N., Berg, A., Lutsko, N.J., Dufour, A., Zeng, Z., Jiang, X, van Dijk, A.I.J.M. & Miralles, D.G. 2023. High-resolution (1 km) Köppen-Geiger maps for 1901–2099 based on constrained CMIP6 projections. Scientific Data 10: 724. DOI: 10.1038/s41597-023-02549-6
BirdLife International. 2019. Hirundo atrocaerulea (amended version of 2016 assessment). The IUCN Red List of Threatened Species: e.T22712318A155549636. DOI: https://dx.doi.org/10.2305/IUCN.UK.2019-3.RLTS.T22712318A155549636.en
Bonachela J.A., Pringle, R.M., Sheffer, E., Coverdale, T.C., Guyton, J.A., Caylor, K.C., Levin, S.A. & Tarnita, C.E. 2015. Termite mounds can increase the robustness of dryland ecosystems to climatic change. Science 347: 651–655. DOI: 10.1126/science.1261487
Bothma, J.D.P. & Le Riche, E.A.N. 1984. Aspects of the ecology and the behaviour of the Leopard Panthera pardus in the Kalahari Desert. Koedoe 27: 259–279. DOI: 10.4102/koedoe.v27i2.585
Braack, L.E.O., 1995. Seasonal activity of savanna termites during and after severe drought. Koedoe 38: 73–82. DOI: 10.4102/koedoe.v38i1.307
Carpaneto, G.M. & Germi, F.P. 1989. The mammals in the zoological culture of the Mbuti pygmies in north-eastern Zaire. Hystrix – Italian Journal of Mammalogy 1: 1–83.
Chambers, J.C. & MacMahon, J.A. 1994. A day in the life of a seed: Movements and fates of seeds and their implications for natural and managed systems. Annual Review of Ecology and Systematics 25: 263-292. 10.1146/annurev.es.25.110194.001403
Chen, A., Hetem, R. & Weyer, N.M. 2023. Aardvarks may be energetically challenged even in a mesic environment – summary of a short study in the Waterberg region. Afrotherian Conservation 18: 2–4.
Cilliers, S. 2002. The ecological importance of the Aardvark. Afrotherian Conservation 1: 7–8.
Couper, L.I., Sanders, N.J., Heller, N.E., & Gordon, D.M. 2021. Multiyear drought exacerbates long-term effects of climate on an invasive ant species. Ecology 102: e03476. DOI: https://doi.org/10.1002/ecy.3476
Davies, A.B., Eggleton, P., van Rensburg, B.J. & Parr, C.L., 2015. Seasonal activity patterns of African savanna termites vary across a rainfall gradient. Insectes Sociaux 62: 157–165. DOI: 10.1007/s00040-014-0386-y
Dean, W.R.J. & Milton, S.J. 1991. Disturbances in semi‐arid shrubland and arid grassland in the Karoo, South Africa: Mammal diggings as germination sites. African Journal of Ecology 29: 11–16.
Deutsch, C.A., Tewksbury, J.J., Huey, R.B., Sheldon, K.S., Ghalambor, C.K., Haak, D.C. & Martin, P.R. 2008. Impacts of climate warming on terrestrial ectotherms across latitude. Proceedings of the National Academy of Sciences 105: 6668–6672. DOI: 10.1073/pnas.0709472105
Eloff, F.C. 1973. Lion predation in the Kalahari Gemsbok National Park. South African Journal of Wildlife Research 3: 59–63. DOI: 10.10520/AJA03794369_2816
Epps, C.W., Weldy, M.J., Crowhurst, R.S. & Spaan, R.S., 2021. Estimating the distribution and habitat suitability for aardvarks (Orycteropus afer) in Kruger National Park, South Africa. African Journal of Ecology 59: 854–865. DOI: 10.1111/aje.12916
Epps, C.W., Crowhurst, R.S., Spaan, R., Weldy, M. & Tavalire, H.F. 2024. Influence of climate and landscape on genetic differentiation of Aardvarks (Orycteropus afer). Diversity and Distributions 30: e13792. DOI: 10.1111/ddi.13792
Evans, S.W. & Bouwman, H. 2010. Habitat selection by blue swallows Hirundo atrocaerulea Sundevall, 1850 breeding in South Africa and its implications for conservation. African Journal of Ecology 48: 871–879. DOI: 10.1111/j.1365-2028.2009.01183.x
Evans, S.W. 2015. Blue Swallow Hirundo atrocaerulea. In: Taylor M.R., Peacock F., Wanless R.M., editors. The 2015 Eskom Red Data book of birds of South Africa, Lesotho and Swaziland. BirdLife South Africa, Johannesburg. pp 48–51. https://speciesstatus.sanbi.org/assessment/last-assessment/3076/
Evans, T., Dawes, T., Ward, P. & Lo, N. 2011. Ants and termites increase crop yield in a dry climate. Nature Communications 2: 262. DOI: 10.1038/ncomms1257
Farji-Brener, A.G., Dalton, M.C., Balza, U., Courtis, A., Lemus-Domínguez, I., Fernández-Hilario, R. & Cáceres-Levi, D. 2018. Working in the rain? Why leaf-cutting ants stop foraging when it’s raining. Insectes Sociaux 65: 233–239. DOI: https://doi.org/10.1007/s00040-018-0605-z
Foster, I.B., McIntyre, T. & Haussmann, N.S. 2022. Understanding the relationship between farmers and burrowing mammals on South African farms: are burrowers friends or foes?. Pp. 69–81. In: Kerr RB, Pendergrast TL, Smith BJ, Liebert J, Eds. Rethinking Food System Transformation. Springer, New York. DOI: 10.1007/s10460-019-09939-w
Fuller, A., Hetem, R.S., Maloney, S.K. & Mitchell, D. 2014. Adaptation to heat and water shortage in large, arid-zone mammals. Physiology 29: 159–167. DOI: 10.1152/physiol.00049.2013
Geerts, S., van der Linden, J. & van der Linden, E. 2016. The ecology and foraging behaviour of the harvester termite, Baucaliotermes hainesi in semi-arid grasslands in the northwestern interior of South Africa. Insectes Sociaux 63: 457–465. DOI: 10.1007/s00040-016-0488-9
Goldman, C.A. 1986. A review of the management of the Aardvark Orycteropus afer in captivity. International Zoo Yearbook 24/25: 286–294. DOI: 10.1111/j.1748-1090.1985.tb02556.x
Gutiérrez, J.M., R.G. Jones, G.T. Narisma, L.M. Alves, M. Amjad, I.V. Gorodetskaya, M. Grose, N.A.B. Klutse, S. Krakovska, J. Li, D. Martínez-Castro, L.O. Mearns, S.H. Mernild, T. Ngo-Duc, B. van den Hurk & J.-H. Yoon, 2021: Atlas. In Climate Change 2021: The Physical Science Basis. Contribution of Working Group I to the Sixth Assessment Report of the Intergovernmental Panel on Climate Change [Masson-Delmotte, V., P. Zhai, A. Pirani, S.L. Connors, C. Péan, S. Berger, N. Caud, Y. Chen, L. Goldfarb, M.I. Gomis, M. Huang, K. Leitzell, E. Lonnoy, J.B.R. Matthews, T.K. Maycock, T. Waterfield, O. Yelekçi, R. Yu, and B. Zhou (eds.)]. Cambridge University Press, Cambridge, United Kingdom and New York, NY, USA, pp. 1927–2058, DOI: 10.1017/9781009157896.021. Interactive Atlas available from https://interactive-atlas.ipcc.ch/
Halsch, C.A., Shapiro, A.M., Fordyce, J.A., Nice, C.C., Thorne, J.H., Waetjen, D.P. & Forister, M.L. 2021. Insects and Recent Climate Change. Proceedings of the National Academy of Sciences 118: e2002543117. DOI: 10.1073/pnas.2002543117
Haltenorth, T. & Diller, H. 1977. Säugetiere Afrikas und Madagaskars. BLV Buchverlag, Munich, Germany. (A field guide to the mammals of Africa including Madagascar. Translation from German by R. W. Hayman for Collins, London, 1980, 400 pp.)
Haussmann, N.S., Louw, M.A., Lewis, S., Nicol, K.J., Van der Merwe, S., & Le Roux, P.C. 2018. Ecosystem engineering through aardvark (Orycteropus afer) burrowing: Mechanisms and effects. Ecological Engineering 118: 66-72. DOI: 10.1016/j.ecoleng.2018.04.022
Hayward, M.W., Henschel, P., O’Brien, J., Hofmeyr, M., Balme, G. & Kerley G.I.H. 2006. Prey preferences of the Leopard (Panthera pardus). Journal of Zoology 270: 298–313. DOI: 10.1111/j.1469-7998.2006.00139.x
Hetem, R.S., Maloney, S.K., Fuller, A. & Mitchell, D. 2016. Heterothermy in large mammals: Inevitable or implemented? Biological Reviews 91: 187–205. DOI: 10.1111/brv.12166
IUCN. 2024. Guidelines for Using the IUCN Red List Categories and Criteria: Version 16. Prepared by the IUCN Standards and Petitions Committee. Downloadable from https://www.iucnredlist.org/documents/RedListGuidelines.pdf
IUCN. 2012. IUCN Red List Categories and Criteria: Version 3.1, second edition. IUCN, Gland, Switzerland and Cambridge, UK. Available at https://www.iucnredlist.org/technical-documents/categories-and-criteria
Johnson, S.D., Eichhoff, A. & Midgley, J.J. 2025. Antbears and underground melons: A highly specialized seed dispersal mutualism mediated by scent. Plants, People, Planet: 1–10. DOI: 10.1002/ppp3.10638
Kingdon J. 1997. The Kingdon field guide to African mammals. 2nd ed. Academic Press. London, UK.
Klarmann, S. 2025. Dead wild animals by numbers — Professional Hunters Association stats tell a grim tale. Daily Maverick, 08 April 2025. https://www.dailymaverick.co.za/opinionista/2025-04-08-dead-wild-animals-by-numbers-professional-hunters-association-stats-tell-a-grim-tale/
Knöthig, J. 2005. Biology of the aardvark (Orycteropus afer). M.Sc. Thesis. Ruprecht-Karls-Universität, Heidelberg, Germany.
Koutchédi, M., Drouilly, M., Lehmann, T. & Nobimè, G. 2023. Insights into the population trends, conservation status and threats for aardvark populations in protected areas in sub-Saharan Africa. Afrotherian Conservation 18: 7–11.
Koutchédi, M. & Nobimè, G. 2021. Anthropogenic pressures on the aardvark (Orycteropus afer) in Monts Kouffé Protected Forest in Benin. Afrotherian Conservation 17: 2–6.
Lawal, S., Sitch, S., Lombardozzi, D., Nabel, J.E.M.S., Wey, H.-W., Friedlingstein, P., Tian, H. & Hewitson, B. 2022. Investigating the response of leaf area index to droughts in southern African vegetation using observations and model simulations. Hydrology and Earth System Sciences 26: 2045–2071. DOI: 10.5194/hess-26-2045-2022
Le Gros Clark, W.E. & Sonntag, C.F. 1926. A monograph of Orycteropus afer. – III. The skull. The skeleton of the trunk and limbs. Proceedings of the Zoological Society, London 30: 445–485.
Lehmann, T. 2007. Amended taxonomy of the order Tubulidentata (Mammalia, Eutheria). Annals of the Transvaal Museum 44: 179–196. DOI: 10.10520/EJC83654
Lehmann, T. 2009. Phylogeny and systematics of the Orycteropodidae (Mammalia, Tubulidentata). Zoological Journal of the Linnean Society 155: 649–702. DOI: 10.1111/j.1096-3642.2008.00460.x
Lindsey, P.A. 1999. The feeding ecology and habitat use of the aardvark Orycteropus afer. M.Sc. Thesis. University of Pretoria, South Africa.
Lindsey, P.A., Balme, G., Becker, M., Begg, C., Bento, C., Bocchino, C., Dickman, A., Diggle, R.W., Eves, H., Henschel, P., Lewis, D., Marnewick, K., Mattheus, J., McNutt, J.W., McRobb, R., Midlane, N., Milanzi, J., Morley, R., Murphree, M., Opyene, V., Phadima, J., Purchase, G., Rentsch, D., Roche, C., Shaw, J., Van der Westhuizen, H., Van Vliet, N. & Zisadza-Gandiwa, P. 2013. The bushmeat trade in African savannas: Impacts, drivers, and possible solutions. Biological Conservation 160: 80–96. DOI: 10.1016/j.biocon.2012.12.020
Linley, G.D., Geary, W.L., Jolly, C.J., Spencer, E.E., Ashman, K.R. Michael, D.R., Westaway, D.M. & Nimmo, D.G. 2024. Wombat burrows are hotspots for small vertebrates in a landscape subject to gigafire. Journal of Mammalogy 105: 752–764. DOI: 10.1093/jmammal/gyae034
Lynch, C.D. 1994. The mammals of Lesotho. Navorsinge van die Nasionale Museum Bloemfontein 10: 177–241.
Makwati, N., Tsvuura, Z. & Kraai, M. 2024. Positive perceptions about the Aardvark (Orycteropus afer) are inadequate to protect the species in northern KwaZulu-Natal, South Africa. Human Dimensions of Wildlife 29: 284–299. DOI: 10.1080/10871209.2023.2234386
Manatsa, D., Chingombe, W., Matsikwa, H. & Matarira, C.H. 2008. The superior influence of Darwin sea level pressure anomalies over ENSO as a simple drought predictor for Southern Africa. Theoretical and Applied Climatology 92: 1–14. DOI: 10.1007/s00704-007-0315-3
Manatsa, D., Mushore, T. & Lenouo, A. 2017. Improved predictability of droughts over Southern Africa using the Standardized Precipitation Evapotranspiration Index and ENSO. Theoretical and Applied Climatology 127: 259–274. DOI: 10.1007/s00704-015-1632-6
Marsh, A.C. 1987. The foraging ecology of two Namib Desert harvester ant species. South African Journal of Zoology 22, 130–136. 10.1080/02541858.1987.11448033
Martin, S.A. 2017. The aardvark as an ecological engineer in the Eastern Karoo: dig patterns and emergent processes. M.Sc. Thesis, Nelson Mandela University, Gqeberha, South Africa.
McNab, B.K. 1984. Physiological convergence amongst anteating and termite-eating mammals. Journal of Zoology 203: 485–510. DOI: 10.1111/j.1469-7998.1984.tb02345.x
Meester, J.A.J., Rautenbach, I.L., Dippenaar, N.J. & Baker, C.M. 1986. Classification of Southern African Mammals. Monograph number 5. Transvaal Museum, Pretoria, South Africa. DOI: 10.10520/AJA090799001_105
Meeuse, A.J.D. 1963. A possible case of interdependence between a mammal and a higher plant. Archive Néerlandaise de Zoologie 13: 314–318.
Melton, D.A. 1976. The biology of Aardvark (Tubulidentata-Orycteropodidae). Mammal Review 6: 75–88. DOI: 10.1111/j.1365-2907.1976.tb00204.x
Melton D.A. & Daniels, C. 1986. A note on the ecology of the aardvark Orycteropus afer. South African Journal of Wildlife Research 16: 112–114.
Monadjem, A. 1998. Distributional patterns and conservation status of mammals of Swaziland, southern Africa. Koedoe 41: 45–59. DOI: 10.4102/koedoe.v41i2.252
Nagy, K., Girard, I.A., & Brown, T.K., 1999. Energetics of free-ranging mammals, reptiles, and birds. Annual Review of Nutrition 19: 247–77. DOI: 10.1146/annurev.nutr.19.1.247
Nel, J.J.C. & Hewitt, P.H. 1969. A study of the food eaten by a field population of the harvester termite, Hodotermes mossambicus (Hagen) and its relation to population density. Journal of the Entomological Society of Southern Africa, 32: 123–131.
Nel, P.J., Taylor, W.A., Meltzer, D.G.A. & Haupt, M.A. 2000. Capture and immobilisation of Aardvark (Orycteropus afer) using different drug combinations. Journal of the South African Veterinary Association 71: 58–63. DOI: 10.4102/jsava.v71i1.679
Nicolai, N., Smeins, F. & Cook, J. 2008. Harvester ant nests improve recovery performance of drought impacted vegetation in grazing regimes of semiarid savanna, Texas. The American Midland Naturalist 160: 29–40. DOI: 10.1674/0003-0031(2008)160[29:HANIRP]2.0.CO;2
Nieman, W.A. 2018. Culture, Conflict, and Cuisine: A quantitative assessment of terrestrial vertebrate off-take at the human wildlife interface. M.Sc. Thesis, University of Stellenbosch, Stellenbosch, South Africa.
Nieman, W.A., Leslie, A.J. & Wilkinson, A. 2019. Traditional medicinal animal use by Xhosa and Sotho communities in the Western Cape Province, South Africa. Journal of Ethnobiology and Ethnomedicine 15: 1–14. DOI: 10.1186/s13002-019-0311-6
Nuttall, P.A. 2022. Climate change impacts on ticks and tick-borne infections. Biologia 77: 1503–1512. DOI: 10.1007/s11756-021-00927-2
Ohiagu, C.E., 1979. A quantitative study of seasonal foraging by the grass harvesting termite, Trinervitermes geminatus (Wasmann), (Isoptera, Nasutitermitinae) in Southern Guinea savanna, Mokwa, Nigeria. Oecologia 40: 179–188. DOI: 10.1007/BF00347935
Pacifici, M., Santini, L., Di Marco, M., Baisero, D., Francucci, L., Grottolo Marasini, G., Visconti, P. & Rondinini, C. 2013. Generation length for mammals. Nature Conservation 5: 87–94. DOI: 10.3897/natureconservation.5.5734
Parmesan, C. 2006. Ecological and evolutionary responses to recent climate change. Annual Review of Ecology and Systematics 37: 637–669. DOI: 10.1146/annurev.ecolsys.37.091305.110100
Parys, A., Lehmann, T., Schoo, W. & Wilms, T.M. 2012. Newcomers enrich the European zoo Aardvark population. Afrotherian Conservation 9: 2–5.
Phakoago, M.V. 2024. Myrmecophagous mammals in a changing world: The ecology of Aardvarks and Temminck’s Pangolins in the Kalahari. Doctoral Dissertation. University of the Witwatersrand, Johannesburg, South Africa.
Phakoago, M.V., Maloney, S.K., Kamerman, P.R., Meyer, L.C.R., Weyer, N.M. & Fuller, A. 2024. Social media as a tool to understand the distribution and ecology of elusive mammals. Journal of Mammalogy 105: 206–214. DOI: 10.1093/jmammal/gyad114
Pike, D.A. & Mitchell, J.C., 2013. Burrow-dwelling ecosystem engineers provide thermal refugia throughout the landscape. Animal Conservation 16: 694–703. DOI: 10.1111/acv.12049
Pocock, R.I. 1924. Some external characters of Orycteropus afer. Proceedings of the Zoological Society, London 94: 697–706.
Pohlová, L., Schepsky, P., Lehmann, T., Hochkirch, A., Masopustová, R., Šimek, J., W. Schoo, R. Vodička, & Robovský, J. 2014. Defining management units for European captive aardvarks. Zoo Biology 33: 433-439. DOI: 10.1002/zoo.21164
Power, R.J. 2002. Prey selection of lions Panthera leo in a small, enclosed reserve. Koedoe 45: 67–75. DOI: 10.4102/koedoe.v45i2.32
Power, R.J. 2014. The distribution and status of mammals in the North West Province. Department of Economic Development, Environment, Conservation & Tourism, North West Provincial Government, Mahikeng, South Africa.
Power, R.J., Van Straaten, A., Schaller, R., Mooke, M., Boshoff, T. & Nel, H.P. 2019. An inventory of mammals of the North West province, South Africa. Annals of the Ditsong National Museum of Natural History 8: 6-29. DOI: 10.10520/EJC-141b4d1b3c
Radloff, F.G. & Du Toit, J.T. 2004. Large predators and their prey in a southern African savanna: a predator’s size determines its prey size range. Journal of Animal Ecology 73: 410–423. DOI: 10.1111/j.0021-8790.2004.00817.x
Ramesh, T. & Downs, C.T. 2015. Impact of land use on occupancy and abundance of terrestrial mammals in the Drakensberg Midlands, South Africa. Journal for Nature Conservation 23: 9–18. DOI: 10.1016/j.jnc.2014.12.001
Rey, B., Costello, M.-A., Fuller, A., Haw, A., Hetem, R.S., Mitchell, D. & Meyer, L.C.R. 2014. Chemical immobilization and anesthesia of free-living Aardvarks (Orycteropus afer) with ketamine-medetomidine-midazolam and isoflurane. Journal of Wildlife Diseases 50: 864–872. DOI: 10.7589/2013-07-166
Rey, B., Fuller, A., Mitchell, D., Meyer, L.C.R. & Hetem, R.S. 2017. Drought-induced starvation of Aardvarks in the Kalahari: An indirect effect of climate change. Biology Letters 13: 1–5. DOI: 10.1098/rsbl.2017.0301
Richard, Y., Fauchereau, N., Poccard, I., Rouault, M. & Trzaska, S. 2001. 20th Century droughts in Southern Africa: Spatial and temporal variability, teleconnections with oceanic and atmospheric conditions. International Journal of Climatology 21: 873–885. DOI: 10.1002/joc.656
Robinson, N.M., Leonard, S.W.J., Ritchie, E.G., Bassett, M., Chia, E.K., Buckingham, S., Gibb, H., Bennett, A.F. & Clarke, M.F. 2013. Refuges for fauna in fire-prone landscapes: their ecological function and importance. Journal of Applied Ecology 50: 1321–1329. DOI: 10.1111/1365-2664.12153
Rouault, M. & Richard, Y. 2005. Intensity and spatial extent of droughts in Southern Africa. Geophysical Research Letters 32: 2–5. DOI: 10.1029/2005GL022436
Roxburgh, D.J. 2010. Prey and range use of lions on Tswalu Kalahari Reserve. M.Sc. Thesis. University of Pretoria, Pretoria, South Africa.
Rust, N.A., Nghikembua, M.T., Kasser, J.J.W. & Marker, L.L. 2015. Environmental factors affect swing gates as a barrier to large carnivores entering game farms. African Journal of Ecology 53: 339–345. DOI: 10.1111/aje.12188
Sarukhán, J. & Whyte, A. 2005. Millennium Ecosystem Assessment. Ecosystems and Human Well-Being: Synthesis. Washington DC: Island Press.
Schumann, M., Schumann, B., Dickman, A., Watson, L.H. & Marker L. 2006. Assessing the use of swing gates in game fences as a potential non-lethal predator exclusion technique. South African Journal of Wildlife Research 36: 173–181. https://hdl.handle.net/10520/EJC117243
Shoshani, J., Goldman, C.A. & Thewissen, J.G.M. 1988. Orycteropus afer. Mammalian Species 300: 1–8. DOI: 10.2307/3503996
Simelane T.S. & Kerley G.I.H. 1998. Conservation implications of the use of vertebrates by Xhosa traditional healers in South Africa. South African Journal of Wildlife Research 28: 121–126. https://hdl.handle.net/10520/EJC117057
Skinner, J.D. & Chimimba, C.T. (eds). 2005. The Mammals of the Southern African Subregion. Cambridge University Press, United Kingdom, Cambridge. DOI: 10.1017/CBO9781107340992
Stuart, C. & Stuart, T. 2001. Field Guide to Mammals of Southern Africa. Struik Publishers, Cape Town, South Africa.
Sundaram, M., Steiner, E., & Gordon, D.M. 2022. Rainfall, neighbors, and foraging: The dynamics of a population of red harvester ant colonies 1988–2019. Ecological Monographs 92: e1503. DOI: 10.1002/ecm.1503
Tabuce, R., Asher, R.J. & Lehmann, T. 2008. Afrotherian mammals: a review of current data. Mammalia 72: 2–14. DOI: 10.1515/mamm.2008.004
Taylor, W.A. 1998. The ecology of the aardvark, Orycteropus afer (Tubulidentata – Orycteropodidae). M.Sc. Thesis. University of Pretoria, Pretoria, South Africa.
Taylor, W.A. 2005. Order Tubulidentata. In J.D. Skinner & C.T. Chimimba (eds.), The Mammals of the Southern African Subregion. Cambridge University Press, Cambridge, UK.
Taylor, W.A. 2013. Orycteropus afer Aardvark. In: J.S. Kingdon, D.H.D. Happold, M. Hoffmann, T. Butynski, M. Happold and J. Kalina (eds), Mammals of Africa. Volume I. Introductory Chapters and Afrotheria, pp. 290–295. Bloomsbury Publishing, London. DOI: 10.1111/aje.12136
Taylor, W.A., Child, M.F., Lindsey, P.A., Nicholson, S.K., Relton, C. & Davies-Mostert, H.T. 2021. South Africa’s private wildlife ranches protect globally significant populations of wild ungulates. Biodiversity & Conservation 30: 4111–4135. DOI: 10.1007/s10531-021-02294-5
Taylor, W.A., Cilliers, S., Meyer, L.C.R. & Wilson, A.-L. 2016. Orycteropus afer. In Child M.F., Roxburgh L., Do Linh San E., Raimondo D., Davies-Mostert H.T., editors. The Red List of Mammals of South Africa, Swaziland and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa. DOI: 10.13140/RG.2.2.11970.68806
Taylor, W.A. & Lehmann, T. 2015. Orycteropus afer. The IUCN Red List of Threatened Species: e.T41504A21286437. DOI: https://dx.doi.org/10.2305/IUCN.UK.2015-2.RLTS.T41504A21286437.en
Taylor, W.A., Lehmann, T. & Weyer, N.M. 2018. Will aardvarks go thirsty under climate change?. Afrotherian Conservation 14: 3–9.
Taylor, W.A., Lehmann, T. & Weyer, N.M. 2019. A roadmap for future Aardvark research. Afrotherian Conservation 15: 25–30.
Taylor, W.A., Lindsey, P.A. & Skinner, J.D. 2002. The feeding ecology of the Aardvark Orycteropus afer. Journal of Arid Environments 50: 135–152. DOI: 10.1006/jare.2001.0854
Taylor, W.A. & Skinner, J.D. 2000. Associative feeding between Aardwolves (Proteles cristatus) and Aardvarks (Orycteropus afer). Mammal Review 30: 141–143. DOI: 10.1046/j.1365-2907.2000.00062.x
Taylor, W.A. & Skinner, J.D. 2001. Associative feeding between Ant-eating Chats, Myrmecocichla formicivora, and Aardvarks, Orycteropus afer. Ostrich 72: 199–200. DOI: 10.2989/00306520109485317
Taylor, W.A. & Skinner, J.D. 2003. Activity patterns, home ranges and burrow use of Aardvarks (Orycteropus afer) in the Karoo. Journal of Zoology 261: 291–297. DOI: 10.1017/S0952836903004217
Taylor, W.A. & Skinner, J.D. 2004. Adaptations of the Aardvark for survival in the Karoo: a review. Transactions of the Royal Society of South Africa 59: 105–108. DOI: 10.1080/00359190409519169
Van Aarde, R.J., Willis, C.K., Skinner, J.D. & Haupt, M.A. 1992. Range utilization by the Aardvark, Orycteropus afer (Pallas, 1766) in the Karoo, South Africa. Journal of Arid Environments 22: 387–394. DOI: 10.1016/S0140-1963(18)30581-0
Van der Vinne, V., Gorter, J.A., Riede, S.J., & Hut, R.A. 2015. Diurnality as an energy-saving strategy: energetic consequences of temporal niche switching in small mammals. The Journal of Experimental Biology 218: 2585-2593. DOI: 10.1242/jeb.119354
Vernon, C.J. & Dean, W.R.J. 1988. Short notes: Further African bird–mammal feeding associations. Ostrich 59: 38–39. 10.1080/00306525.1988.9633925
Waddell, P.J., Kishino, H. & Ota, R. 2001. A phylogenetic foundation for comparative mammalian genomics. Genome Informatics 12: 141-154.
Wakelin, J. & Hill, T.R. 2007. The impact of land transformation on breeding Blue Swallows Hirundo atrocaerulea Sundevall, in KwaZulu-Natal, South Africa. Journal for Nature Conservation 15: 245–255. DOI 10.1016/j.jnc.2007.05.003
Wakelin, J., Wilson, A.-L. & Downs, C.T. 2013. Ground cavity nest temperatures and their relevance to Blue swallow, Hirundo atrocaerulea, conservation. Ostrich 84: 221–226. DOI: 10.2989/00306525.2013.859640
Weise, F.J., Wessels, Q., Munro, S. & Solberg, M. 2014. Using artificial passageways to facilitate the movement of wildlife on Namibian farmland. South African Journal of Wildlife Research 44: 161–166. DOI: 10.3957/056.044.0213
Weyer, N.M., 2018. Physiological flexibility of free-living Aardvarks (Orycteropus afer) in response to environmental fluctuations. Doctoral Dissertation. University of the Witwatersrand, Johannesburg, South Africa. https://hdl.handle.net/10539/25334
Weyer, N.M., Fuller, A., Haw, A.J., Meyer, L.C.R., Mitchell, D., Picker, M., Rey, B. & Hetem, R.S., 2020a. Increased diurnal activity is indicative of energy deficit in a nocturnal mammal, the Aardvark. Frontiers in Physiology 11: 1–15. DOI: 10.3389/fphys.2020.00637
Weyer, N.M., Taylor, A. & Lehmann, T. 2020b. Demystifying Aardvark tracks and how (not) to determine Aardvark sex. Afrotherian Conservation 16: 4–7.
Whitford, W.G. & Kay, F.R. 1999. Biopedturbation by mammals in deserts: a review. Journal of Arid Environments 41: 203–230. DOI: 10.1006/jare.1998.0482
Whiting, M.J., Williams, V.L. & Hibbitts, T.J. 2011. Animals traded for traditional medicine at the Faraday market in South Africa: Species diversity and conservation implications. Journal of Zoology 284: 84–96. DOI: 10.1111/j.1469-7998.2010.00784.x
Whittington-Jones, G.M. 2006. The role of Aardvarks (Orycteropus afer) as ecosystem engineers in arid and semi-arid landscapes of South Africa. M.Sc. Thesis. Rhodes University, Grahamstown, South Africa.
Whittington-Jones, G.M., Bernard, R.T. & Parker, D.M. 2011. Aardvark burrows: a potential resource for animals in arid and semi-arid environments. African Zoology 46: 362–370. DOI: 10.3377/004.046.0215
Williams, K.S., Williams, S.T., Fitzgerald, L.E., Sheppard, E.C. & Hill, R.A. 2018. Brown Hyaena and Leopard diets on private land in the Soutpansberg Mountains, South Africa. African Journal of Ecology 56: 1021–1027. DOI: 10.1111/aje.12539
Willis C.K.R., Skinner J.D. & Robertson H.G. 1992. Abundance of ants and termites in the False Karoo and their importance in the diet of the aardvark Orycteropus afer. African Journal of Ecology 30: 322–334. DOI: 10.1111/j.1365-2028.1992.tb00509.x
Woodgate Z., Drouilly, M., Distiller, G. & O’Riain, M.J. 2023. The effect of multi-use landscapes on mammal assemblages and its implication for conservation. Land 12: 599. DOI: 10.3390/land12030599
Yalden, D.W., Largen, M.J., Kock, D. & Hillman, J.C. 1996. Catalogue of the Mammals of Ethiopia and Eritrea. 7. Revised checklist, zoogeography and conservation. Tropical Zoology 9: 73-164. DOI: 10.1080/03946975.1996.10539304
Yuan, X., Wang, L. & Wood, E.F. 2018. Anthropogenic intensification of Southern African flash droughts as exemplified by the 2015/16 season. Bulletin of the American Meteorological Society 99: S86–S90. DOI: 10.1175/BAMS-D-17-0077.1